
Each of the following names is wrong. Give the structure and correct name of
each compound.
a
b
c.
d.
(a)
Interpretation:
The structural formula of
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.11E
The structural formula of
The correct name is
Explanation of Solution
The given name is

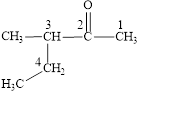
Figure 1
The longest carbon chain contains five carbon atoms. One methyl substituent is attached to the third carbon atom. The functional group present in the given compound is ketone
The structural formula is shown in Figure 1.
The correct name of the compound is
(b)
Interpretation:
The structural formula of
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.11E
The structural formula of
The correct name is
Explanation of Solution
The given name is

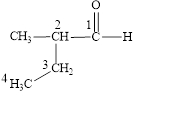
Figure 2
The longest carbon chain contains four carbon atoms. One methyl substituent is attached to the second carbon atom. The functional group present in the given compound is aldehyde
The structural formula is shown in Figure 2.
The correct name of the compound is
(c)
Interpretation:
The structural formula of
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.11E
The structural formula of
The correct name is
Explanation of Solution
The given name is

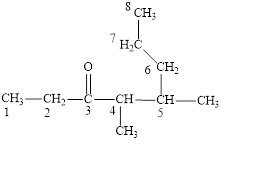
Figure 3
The longest carbon chain contains eight carbon atoms. One methyl substituent is attached to the fourth carbon atom and the other methyl group is attached to the fifth carbon atom. The functional group present in the given compound is ketone
The structural formula is shown in Figure 3.
The correct name of the compound is
(d)
Interpretation:
The structural formula of
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.11E
The structural formula of
The correct name is
Explanation of Solution
The given name is

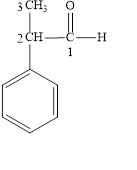
Figure 4
The longest carbon chain contains three carbon atoms. One phenyl substituent is attached to the second carbon atom. The functional group present in the given compound is aldehyde
The structural formula is shown in Figure 4.
The correct name of the compound is
Want to see more full solutions like this?
Chapter 14 Solutions
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
- Which of the following molecules gives a condensation reaction with benzaldehyde? A. 4-methylbenzaldehyde B. 4-chlorobenzaldehyde C. 2,2-methyl propanoic acid D. 3-methylbutanoic acid E. 3-methylbenzaldehydearrow_forward21. Which is a correct IUPAC name for the following CH₁ CH₂ CH₂-CH-CH₂-OH A. 3-methyl-1-hexanol B. 2-ethyl-2-pentanol C. 2-ethyl-1-pentanol D. I-ethyl-2-pentanol CH₂arrow_forwardWhich of the following carboxylic acid is toxic when ingested? a.Citric acid b.Malic acid c.Acetic acid d.Oxalic acid What is the general formula for Grignard reagents? a.RCOOH b.RMgX c.RX d.RCOX Oxidation of alkyl benzenes in the presence of Sulfuric acid will produce which carboxylic acid? a.Cyclohexanecarboxylic acid b.Benzoic acid c.Acrylic acid d.Fumaric acid What dicarboxylic acid contains six carbon atoms in its structure? a.Capric acid b.Pimelic acid c.Adipic acid d.Hexanoic acid Which of the following is an example of an unsaturated carboxylic acid? a.Succinic acid b.Formic acid c.Glycolic acid d.Acrylic acidarrow_forward
- What is the systematic IUPAC name for the given compound? CH3 CH3 CH;CHCH,CH,CH2 -Ń-CH3 a. N-methyl-4-methylhexan-1-amine b. 2,4-dimethylhexan-1-amine c. 2,2,N-trimethylpentan-1-amine d. N,N,4-trimethylpentan-1-aminearrow_forwardIsomer of 2-hexenoic acid A.) Propyl propanoate B.) Hexenoic anhydride C.) Benzoic acid D.) 2-cyclopropyl-propanoic acid E.) None of the given choicesarrow_forwardGive the IUPAC name of the following compound он Он a. 3-ethyl-4,5-hexanediol b. 4-ethyl-2,3-heptanediol c. 3-ethyl-4,5-heptanediol O d. 4-ethyl-2,3-hexanediolarrow_forward
- A compound named 5-(1- Cyanoethy)-2-methylhexanoic acid has something wrong according to IUPAC. What is the correct IUPAC name? O A. 5-Ethylcyanide-2-methylhexanoic acid B. 6-Cyano-2,5-dimethylheptanoic acid C. 6-Cyano-5-methyl-2-heptanoic acid D. 6-Carboxy-2,3-dimethylheptane nitrilearrow_forward1. Which of the following statements is true? I. Aldehydes and ketones both contain a hydroxyl group. II. The names for aldehydes and ketones are derived from the name of the longest carbon chain that contains the carbonyl group. III. The aldehyde and ketone with a molecular formula of C3H6O are constitutional isomers. IV. 2-Propanone is immiscible in water. A. I & II B. II & II C. I & III D. I & IV 2. Whicb of the following is the correct IUPAC name of the structure below?arrow_forwardWrite the structure formulas for each of the following compounds.i) 2-Chloro-pentanoic acid ii) 2-isopropyl-5-methylphenol iii) Ethyl cyclopentyl etheriv) Cyclohexanone v) 2,2-Dimethylpropanalarrow_forward
- Draw the structure of the 2-phenylpentane.arrow_forwardWhich of the following compounds in the simplest ketone? A.) methanone B.) ethanone C.) propanone D.) butanone E.) pentanonearrow_forwardWhich of the following is the correct IUPAC name for the following compound? A. 3-hydroxo-3-methylpentanal B. 3-hydroxyl-3-methylpentanal C. 3-hydroxyl-3-methylpentanone D. 3-hydroxyl-4-methylpentanal E. none of the abovearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





