
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.6, Problem 18P
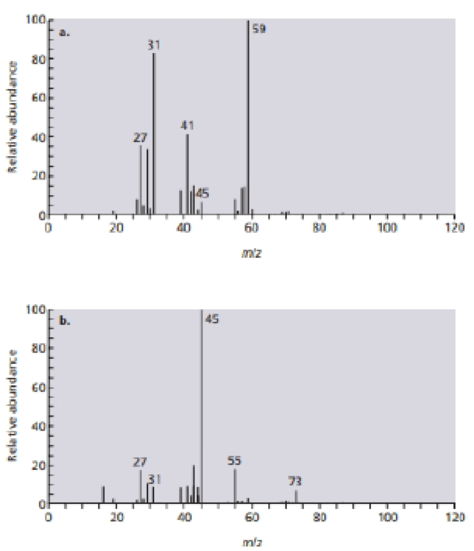
The reaction of (Z)-2-pentene with water and a trace of H2SO4 forms two products. Identity the products from their mass spectra
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Arrange the alkenes in each set in order of increasing rate of reaction with HI and explain the basis for your ranking. Draw the structural formula of the major product formed in each case.
Identify products A and B from the given 1H NMR data.
Treatment of acetone [(CH3)2C=O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1H) ppm. What is the structure of B?
Identify products A and B from the given 1H NMR data.
Treatment of CH2=CHCOCH3 with one equivalent of HCl forms compound A. A exhibits the following absorptions in its 1H NMR spectrum: 2.2 (singlet, 3H), 3.05 (triplet, 2 H), and 3.6 (triplet, 2 H) ppm. What is the structure of A?
Chapter 13 Solutions
Organic Chemistry (8th Edition)
Ch. 13.1 - Which of the following fragments produced in a...Ch. 13.2 - What distinguishes the mass spectrum of...Ch. 13.2 - What is the most likely m/z value for the base...Ch. 13.3 - Prob. 5PCh. 13.3 - a. Suggest possible molecular formulas for a...Ch. 13.3 - If a compound has a molecular ion with an...Ch. 13.3 - Identify the hydrocarbon that has a molecular ion...Ch. 13.4 - Predict the relative intensities of the molecular...Ch. 13.5 - Which molecular formula has an exact molecular...Ch. 13.5 - Prob. 11P
Ch. 13.6 - Sketch the mass spectrum expected for...Ch. 13.6 - The mass spectra of 1-methoxybutane,...Ch. 13.6 - Primary alcohols have a strong peak at m/z = 31....Ch. 13.6 - Identify the ketones responsible for the mass...Ch. 13.6 - Prob. 16PCh. 13.6 - Using curved arrows, show the principal fragments...Ch. 13.6 - The reaction of (Z)-2-pentene with water and a...Ch. 13.9 - a. Which is higher in energy: electromagnetic...Ch. 13.9 - Prob. 20PCh. 13.13 - Prob. 21PCh. 13.14 - Which occur at a larger wavenumber: a. the C O...Ch. 13.14 - Prob. 23PCh. 13.14 - Prob. 24PCh. 13.14 - Rank the following compounds from highest...Ch. 13.14 - Which shows an O H stretch at a larger...Ch. 13.16 - Prob. 27PCh. 13.16 - a. An oxygen-containing compound shows an...Ch. 13.16 - Prob. 29PCh. 13.16 - For each of the following pair of compounds, name...Ch. 13.17 - Which of the following compounds has a vibration...Ch. 13.17 - Prob. 32PCh. 13.18 - A compound with molecular formula C4H6O gives the...Ch. 13.20 - Prob. 34PCh. 13.20 - Prob. 35PCh. 13.21 - Predict the max of the following compound:Ch. 13.21 - Prob. 37PCh. 13.23 - a. At pH = 7 one of the ions shown here is purple...Ch. 13.23 - Prob. 39PCh. 13.23 - Prob. 40PCh. 13 - In the mass spectrum of the following compounds,...Ch. 13 - Prob. 42PCh. 13 - Draw structures for a saturated hydrocarbon that...Ch. 13 - Rank the following compounds in order of...Ch. 13 - For each of the following pairs of compounds,...Ch. 13 - a. How could you use IR spectroscopy to determine...Ch. 13 - Assuming that the force constant is approximately...Ch. 13 - Norlutin and Enovid are ketones that suppress so...Ch. 13 - In the following boxes, list the types of bonds...Ch. 13 - A mass spectrum shows significant peaks at m/z. =...Ch. 13 - Prob. 51PCh. 13 - Prob. 52PCh. 13 - Prob. 53PCh. 13 - The IR spectrum of a compound with molecular...Ch. 13 - Rank the following compounds from highest...Ch. 13 - Rank the following compounds from highest...Ch. 13 - What peaks in their mass spectra can be used to...Ch. 13 - Prob. 58PCh. 13 - Which one of the following five compounds produced...Ch. 13 - Prob. 60PCh. 13 - Each of the IR spectra shown below is accompanied...Ch. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - How can IR spectroscopy distinguish between...Ch. 13 - Prob. 65PCh. 13 - Prob. 66PCh. 13 - Give approximate wavenumbers for the major...Ch. 13 - Prob. 68PCh. 13 - Which one of the following live compounds produced...Ch. 13 - Phenolphthalein is an acid-base indicator. In...Ch. 13 - Prob. 71PCh. 13 - How can you use UV spectroscopy to distinguish...Ch. 13 - Prob. 73PCh. 13 - The IR and mass spectra for three different...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7. What effect would you expect the following changes to have on the rate of the SN2 reaction of 1-iodo-2-methylbutane with cyanide ion? (a) The CN' concentration is halved, and the 1-iodo-2-methylbutane concentration is doubled. (b) Both the CN1 and the 1-iodo-2-methylbutane concentrations are tripled.arrow_forward(ii) Draw the product R, including a detailed reaction mechanism for the conjugate addition reaction between the 1,3-dicarbonyl P and the a,ß-unsaturated ketone Q. Eto P OEt + Q OEt Et3N R (iv) Explain why conjugate addition is favoured over direct addition to Q in this case.arrow_forwardTwo products are obtained from the reaction of (Z)-pent-2-ene with water in sulfuric acid. The mass spectra of these products are shown below. Draw the structure of each product, and match the product to its mass spectrumarrow_forward
- The reaction of methylpropene with HBr, under radical conditions, gives two intermediates. Propose a mechanism for the formation of the two products. Propose a mechanism for the following reaction and use electronic factors to account for the formation of a major product: CH2 CH2Br N-Bromosuccinimide (NBS) ho, CCI4 Draw the structure of an antioxidant, Vitamin E free radical and use resonance structures o account for its stability.arrow_forwardOct-1-yne (HC≡CCH2CH2CH2CH2CH2CH3) reacts rapidty with NaH, forming a gas that bubbles out of the reaction mixture, as one product. Oct-1-yne also reacts rapidty with CH3MgBr, and a different gas is produced. Write balanced equations for both reactions and identify the gases formed.arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs due to a nucleophilic substitution pathway. The reaction is 100% stereospecific. Please explain what this observation tells about the mechanism of the reaction.arrow_forward
- 134. The major product formed in the following reaction sequence is: OMe (1) (CF;CO),O; NE13 (A) (2) O3; Me,S MeOarrow_forwardRank the compounds in each group in order of increasing reactivity in electrophilic aromatic substitution: (a) C6H6, C6H5Cl, C6H5CHO; (b) C6H5CH3, C6H5NH2, C6H5CH2NH2.arrow_forwardThe treatment of (CH3)2C = CHCH2Br with H2O forms B (molecular formula C5H10O) as one of the products. Determine the structure of B from its H NMR and IR spectra.arrow_forward
- Treatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forward2) The following compounds are treated with one molar equivalent of Clz and catalytic FeCl3: H N. NH3 NH2 H a) Rank the compounds according to their relative reactivity under the reaction conditions (1 = most reactive; 5 = least reactive) %3D %3D b) Predict the major product obtained in each reaction (assume only one reaction happens on each aromatic starting material).arrow_forwardCompound A on ozonolysis yields the two products shown. What is the structure of compound A? Compound A 1.03 2. (CH3)2S ||| ol H H || IV H Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY