
Chemistry: Atoms First
3rd Edition
ISBN: 9781259638138
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13.2, Problem 1PPC
Interpretation Introduction
Interpretation:
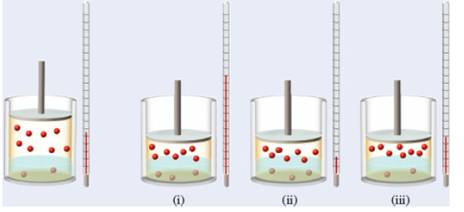
In the following diagram which is (are) represents a closed system when the piston is moved downward, decreasing the volume of the gas over the water has to be determined.
Note: Thermometer indicating the temperature of the system.
Concept introduction:
Henry’s law: Henry’s law states that dissolved gases in liquid are directly proportional to the partial pressure of the gas over the solution. The equation is given by
Where,
c is molar concentration of dissolved gas
k is proportionality constant
P is partial pressure of the gas over the solution
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Suppose you have 600.0 grams of room temperature water (20.0 degrees Celsius) in a thermos. You drop 90.0 grams of ice at 0.00 degrees Celsius into the thermos and shut the lid.(a) What is the equilibrium temperature of the system?
(b) How much ice is left (in grams)?
Provide a step-by-step explanation for how you arrived at your solution as though you were teaching a student to solve this type of problem.
Osmotic pressures are often reported in units of atmospheres or mm Hg. The latter impliesthat the height of a column of liquid can be used as a measure of pressure. This is, in fact, thebasis of the torricellian barometer from which the units of “torr” come. The pressure, P, isrelated to the height of the column by:P = ρghwhere ρ is the density of the liquid, g is the acceleration due to gravity and h is the height. Whatwill be the height (in mm) of a column of aqueous solution with a density of 0.9987 g/mL, if theosmotic pressure of the solution at 25 °C is 1.4 mm Hg? The density of mercury at thistemperature is 13.8 g/mL
What is the mole fraction of potassium permanganate, KMnO4, in an aqueous solution that contains 1.0049 g of potassium permanganate dissolved in 26.2858 grams of water? MW(KMnO4) = 158.034 g/mol.
Chapter 13 Solutions
Chemistry: Atoms First
Ch. 13.2 - Determine for each solute whether the solubility...Ch. 13.2 - Predict whether iodine (I2) is more soluble in...Ch. 13.2 - Prob. 1PPBCh. 13.2 - Prob. 1PPCCh. 13.3 - Prob. 13.2WECh. 13.3 - Determine (a) the molality and (b) the percent by...Ch. 13.3 - Prob. 2PPBCh. 13.3 - Prob. 2PPCCh. 13.3 - Rubbing alcohol is a mixture of isopropyl alcohol...Ch. 13.3 - Prob. 3PPA
Ch. 13.3 - Prob. 3PPBCh. 13.3 - Prob. 3PPCCh. 13.3 - Prob. 13.3.1SRCh. 13.3 - Prob. 13.3.2SRCh. 13.3 - Prob. 13.3.3SRCh. 13.3 - At 25.0C, an aqueous solution that is 25.0 percent...Ch. 13.4 - Calculate the concentration of carbon dioxide in a...Ch. 13.4 - Calculate the concentration of CO2 in water at 25C...Ch. 13.4 - Prob. 4PPBCh. 13.4 - Prob. 4PPCCh. 13.4 - Prob. 13.4.1SRCh. 13.4 - Prob. 13.4.2SRCh. 13.5 - Prob. 13.5WECh. 13.5 - Calculate the vapor pressure of a solution made by...Ch. 13.5 - Calculate the mass of urea that should be...Ch. 13.5 - The diagrams [(i)(iv)] represent four closed...Ch. 13.5 - Ethylene glycol [CH2(OH)CH2(OH)] is a common...Ch. 13.5 - Prob. 6PPACh. 13.5 - What mass of ethylene glycol must be added to 1525...Ch. 13.5 - Prob. 6PPCCh. 13.5 - Prob. 13.7WECh. 13.5 - Prob. 7PPACh. 13.5 - Prob. 7PPBCh. 13.5 - A solution contains 75.0 g of glucose (molar mass...Ch. 13.5 - Prob. 13.5.2SRCh. 13.5 - Prob. 13.5.3SRCh. 13.5 - A 1.00-m solution of HC1 has a freezing point of...Ch. 13.6 - Quinine was the first drug widely used to treat...Ch. 13.6 - Prob. 8PPACh. 13.6 - Prob. 8PPBCh. 13.6 - Prob. 8PPCCh. 13.6 - Prob. 13.9WECh. 13.6 - A solution made by dissolving 25 mg of insulin in...Ch. 13.6 - What mass of insulin must be dissolved in 50.0 mL...Ch. 13.6 - The first diagram represents one aqueous solution...Ch. 13.6 - Prob. 13.10WECh. 13.6 - An aqueous solution that is 0.0100 M in acetic...Ch. 13.6 - Prob. 10PPBCh. 13.6 - Prob. 10PPCCh. 13.6 - Prob. 13.6.1SRCh. 13.6 - A 0.010-M solution of the weak electrolyte HA has...Ch. 13 - Which of the following processes is accompanied by...Ch. 13 - For each of the processes depicted here, determine...Ch. 13 - For each of the processes depicted here, determine...Ch. 13 - Prob. 13.4KSPCh. 13 - Prob. 13.1QPCh. 13 - Prob. 13.2QPCh. 13 - Prob. 13.3QPCh. 13 - Prob. 13.4QPCh. 13 - Prob. 13.5QPCh. 13 - Prob. 13.6QPCh. 13 - Explain why dissolving a solid almost always leads...Ch. 13 - Describe the factors that affect the solubility of...Ch. 13 - Prob. 13.9QPCh. 13 - Prob. 13.10QPCh. 13 - Arrange the following compounds in order of...Ch. 13 - Prob. 13.12QPCh. 13 - Prob. 13.13QPCh. 13 - Prob. 13.14QPCh. 13 - Prob. 13.15QPCh. 13 - Calculate the amount of water (in crams) that must...Ch. 13 - Calculate the molality of each of the following...Ch. 13 - Prob. 13.18QPCh. 13 - Calculate the molalities of the following aqueous...Ch. 13 - For dilute aqueous solutions in which the density...Ch. 13 - Prob. 13.21QPCh. 13 - PepsiCo, maker of Pepsi, announced in April 2015...Ch. 13 - After sales of new Diet Pepsi proved...Ch. 13 - The density of an aqueous solution containing 25.0...Ch. 13 - Prob. 13.25QPCh. 13 - Discuss the factors that influence the solubility...Ch. 13 - What is thermal pollution? Why is it harmful to...Ch. 13 - Prob. 13.28QPCh. 13 - A student is observing two beakers of water. One...Ch. 13 - A man bought a goldfish in a pet shop. Upon...Ch. 13 - The solubility of KNO3 is 155 g per 100 g of water...Ch. 13 - A 3.20-g sample of a salt dissolves in 9.10 g of...Ch. 13 - The solubility of CO2 in water at 25C and 1 atm is...Ch. 13 - Prob. 13.34QPCh. 13 - Fish breathe the dissolved air in water through...Ch. 13 - The solubility of N2 in blood at 37C and at a...Ch. 13 - The difference between water-soluble and...Ch. 13 - Predict whether each vitamin will be water soluble...Ch. 13 - Prob. 13.39QPCh. 13 - The first diagram represents an open system with...Ch. 13 - The diagrams represent an aqueous solution at two...Ch. 13 - Prob. 13.42QPCh. 13 - Prob. 13.43QPCh. 13 - Write the equation representing Raoults law, and...Ch. 13 - Prob. 13.45QPCh. 13 - Write the equations relating boiling-point...Ch. 13 - Prob. 13.47QPCh. 13 - Prob. 13.48QPCh. 13 - What is osmosis? What is a semipermeable membrane?Ch. 13 - Prob. 13.50QPCh. 13 - Prob. 13.51QPCh. 13 - Prob. 13.52QPCh. 13 - What are ion pairs? What effect does ion-pair...Ch. 13 - Prob. 13.54QPCh. 13 - Prob. 13.55QPCh. 13 - Prob. 13.56QPCh. 13 - Prob. 13.57QPCh. 13 - The vapor pressure of benzene is 100.0 mmHg at...Ch. 13 - The vapor pressures of ethanol (C2H5OH) and...Ch. 13 - The vapor pressure of ethanol (C2H5OH) at 20C is...Ch. 13 - Prob. 13.61QPCh. 13 - What arc the boiling point and freezing point of a...Ch. 13 - Prob. 13.63QPCh. 13 - How many liters of the antifreeze ethylene glycol...Ch. 13 - Prob. 13.65QPCh. 13 - Prob. 13.66QPCh. 13 - What are the normal freezing points and boiling...Ch. 13 - At 25C, the vapor pressure of pure water is 23.76...Ch. 13 - Both NaCl and CaCl2 are used to melt ice on roads...Ch. 13 - A 0.86 percent by mass solution of NaCl is called...Ch. 13 - Prob. 13.71QPCh. 13 - Calculate the osmotic pressure of a 0.0500 M MgSO4...Ch. 13 - The tallest trees known are the redwoods in...Ch. 13 - Calculate the difference in osmotic pressure (in...Ch. 13 - Prob. 13.75QPCh. 13 - Consider two aqueous solutions, one of sucrose...Ch. 13 - Arrange the following solutions in order of...Ch. 13 - Prob. 13.78QPCh. 13 - Indicate which compound in each of the following...Ch. 13 - Describe how you would use freezing-point...Ch. 13 - Prob. 13.81QPCh. 13 - The elemental analysis of an organic solid...Ch. 13 - A solution of 2.50 g of a compound having the...Ch. 13 - The molar mass of benzoic acid (C6H5COOH)...Ch. 13 - A solution containing 0.8330 g of a polymer of...Ch. 13 - Prob. 13.86QPCh. 13 - A solution of 6.S5 g of a carbohydrate m 100.0 g...Ch. 13 - A 0.036-M aqueous nitrous acid (HNO2) solution has...Ch. 13 - Prob. 13.89QPCh. 13 - Prob. 13.90QPCh. 13 - Prob. 13.91QPCh. 13 - Prob. 13.92QPCh. 13 - Prob. 13.93QPCh. 13 - Lysozyme is an enzyme that cleaves bacterial cell...Ch. 13 - The blood sugar (glucose) level of a diabetic...Ch. 13 - Trees in cold climates may be subjected to...Ch. 13 - Prob. 13.97QPCh. 13 - Two liquids A and B have vapor pressures of 76 and...Ch. 13 - Determine the van't Hoff factor of Na3PO4 in a...Ch. 13 - Prob. 13.100QPCh. 13 - Consider the three mercury manometers shown in the...Ch. 13 - A forensic chemist is given a white powder for...Ch. 13 - Prob. 13.103QPCh. 13 - A solution of 1.00 g of anhydrous aluminum...Ch. 13 - Explain why reverse osmosis is (theoretically)...Ch. 13 - What masses of sodium chloride, magnesium...Ch. 13 - The osmotic pressure of blood plasma is...Ch. 13 - Prob. 13.108QPCh. 13 - A protein has been isolated as a salt with the...Ch. 13 - A nonvolatile organic compound Z was used to make...Ch. 13 - Prob. 13.111QPCh. 13 - State which of the alcohols listed in Problem...Ch. 13 - Before a carbonated beverage bottle is sealed, it...Ch. 13 - Iodine (I2) is only sparingly soluble in water...Ch. 13 - (a) The root cells of plants contain a solution...Ch. 13 - Hemoglobin, the oxygen-transport protein, binds...Ch. 13 - Prob. 13.117QPCh. 13 - In the apparatus shown, what will happen if the...Ch. 13 - Concentrated hydrochloric acid is usually...Ch. 13 - Explain each of the following statements: (a) The...Ch. 13 - Prob. 13.121QPCh. 13 - A 1.32-g sample of a mixture of cyclohexane...Ch. 13 - How does each of the following affect the...Ch. 13 - A solution contains two volatile liquids A and B....Ch. 13 - Prob. 13.125QPCh. 13 - A mixture of ethanol and 1-propanol behaves...Ch. 13 - Ammonia (NH3) is very soluble in water, but...Ch. 13 - For ideal solutions, the volumes are additive....Ch. 13 - Acetic acid is a weak acid that ionizes in...Ch. 13 - Which vitamins (sec the given structures) do you...Ch. 13 - Calculate the percent by mass of the solute in...Ch. 13 - Acetic acid is a polar molecule and can form...Ch. 13 - Prob. 13.133QPCh. 13 - Fish in the Antarctic Ocean swim in water at about...Ch. 13 - Why are ice cubes (e.g., those you see in the...Ch. 13 - Prob. 13.136QPCh. 13 - Two beakers are placed in a closed container...Ch. 13 - (a) Derive the equation relating the molality (m)...Ch. 13 - At 27C, the vapor pressure of pure water is 23.76...Ch. 13 - A very long pipe is capped at one end with a...Ch. 13 - A mixture of liquids A and B exhibits ideal...Ch. 13 - Use Henrys law and the ideal gas equation to prove...Ch. 13 - Prob. 13.143QPCh. 13 - Prob. 13.144QPCh. 13 - The diagram here shows vapor pressure curves for...Ch. 13 - Valinomycin is an antibiotic. It functions by...Ch. 13 - Prob. 13.147QPCh. 13 - Prob. 13.148QPCh. 13 - Prob. 13.149QPCh. 13 - Explain why we cannot use osmotic pressure to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculate the mass of ethylene glycol (C₂H6O₂) that must be added to 1.00 kg of ethanol (C₂H5OH) to reduce its vapor pressure by 11.0 torr at 35 °C. The vapor pressure of pure ethanol at 35 °C is 1.00 x 10² torr Express the mass in grams to three significant figures. V—| ΑΣΦ m = Submit Previous Answers Request Answer ? X Incorrect; Try Again; 2 attempts remaining garrow_forwardA reaction mixture that consisted of 0.20 mol N2(g) and 0.20 mol H2(g) was introduced into a reactor of volume 25.0 L and heated. At equilibrium, 5.0% of the nitrogen gas had reacted. What is the value of the equilibrium constant KC for the reaction N2(g) + 3H2(g) ⇋ 2 NH3(g) at this temperature?arrow_forwardThe partial molar volumes of two liquids A and B in a mixture in which the mole fraction of A is 0.3713 are 188.2 cm3 mol−1 and 176.14 cm3 mol−1, respectively. The molar masses of the A and B are 241.1 g mol−1 and 198.2 g mol−1. What is the volume of a solution of mass 1.000 kg?arrow_forward
- Determine the mole fraction of acetone in a 1.00 m solution of acetone (CH3COCH3) dissolved in ethanol (CH3CH3OH). The density of acetone is 0.788 g cm–3 and the density of ethanol is 0.789 g cm–3. Enter in decimal form (0.0111, 0.00220, 0.300 etc.)arrow_forwardThe partial pressure of CO2 gas above the liquid in a bottle of champagne at 20°C is 5.5 atm. What is the solubility of CO2 in champagne? Assume Henry’s law constant is the same for champagne as for water: at 20°C, kH = 3.7 x 10-2 mol/L⋅atm.arrow_forwardYou have a flask weighing 121.214 g. To this flask, you added 25.00 mL KCl(aq) solution. You weigh the flask, it is now 150.234 g. You then take the flask to a heating source and evaporate all solvents. When the flask cool down to room temperature, you weigh the flask again, it is 124.231 g. What is the molality of the KCI (aq) solution? The amus for K is 39 and Cl is 35.5, respectivelyarrow_forward
- Calculate the height of a column of water equivalent to 1.00 bar. The density of water is 1.00 g/cm3; the density of mercury is 13.53 g/cm3. Physical Chemistry 1 please provide complete solution.arrow_forward3. (a) The Lattice enthalpy for the solid ionic compound AgBr is +900. kJ/mole. Write the chemical equation that corresponds to the Lattice Enthalpy for AgBr(s) in the space above. Then explain in your own words why this is a large positive number. (b) The hydration enthalpy for AgBr is -821 kJ/mole. Write the chemical equation that corresponds to the Hydration Enthalpy for AgBr(s) in the space above. Then explain in your own words why this is a large negative number.…arrow_forward3. (a) The Lattice enthalpy for the solid ionic compound AgBr is +900. kJ/mole. Write the chemical equation that corresponds to the Lattice Enthalpy for AgBr(s) in the space above. Then explain in your own words why this is a large positive number. (b) The hydration enthalpy for AgBris -821 kJ/mole. Write the chemical equation that corresponds to the Hydration Enthalpy for AgBr(s) in the space above. Then explain in your own words why this is a large negative number. (c) Would you expect this compound to be soluble in water? Why/Why not? Calculate anything you need in order to figure this out, and explain your answer.arrow_forward
- 6 ST4E.1- Calculate the boiling point temperature of phenol (in the Celsius scale) if 4.3 grams of sucrose are dissolved 119 milliliters of phenol. Phenol has a density of 1.07 g/mL, freezing point of 40.5°C, and boiling point of 181.7°C. Type your answer...arrow_forwardCamphor (C10H16O) melts at 179.8 °C, and it has aparticularly large freezing-point-depression constant,Kf = 40.0 °C/m. When 0.186 g of an organic substance of unknownmolar mass is dissolved in 22.01 g of liquid camphor,the freezing point of the mixture is found to be 176.7 °C.What is the molar mass of the solute?arrow_forward5. When 10 g of a certain substance was dissolved in 100 g of water, the boiling point of water became 101.02 ° C. What is the molar mass of this substance? Ebullioscopic elevation Kb = 0.51 K · kg / mol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY