
Concept explainers
(a)
Interpretation:
The reagent needed to convert 2-methylpropene to
Concept Introduction:
A
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
Hydrohalogenation reaction is an addition reaction in which the hydrogen and halogen atoms like Cl, Br are bonded on un-statured carbon atoms of
Answer to Problem 13.67P
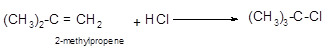
Explanation of Solution
The conversion of 2-methylpropene to
To get the hydrohalogenation product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H-Cl bond of the reagent.
- Add one Cl atom to double-bonded C atom to form new C−Cl single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-X will bond to that double-bonded C atom which has more number of H atoms.
Hence, the hydrohalogenation of 2-methylpropene can be written as follows
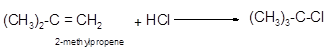
(b)
Interpretation:
The reagent that is needed to convert 2-methylpropene to
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
Hydrohalogenation reaction is an addition reaction in which the hydrogen and halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Answer to Problem 13.67P
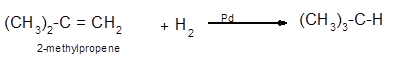
Explanation of Solution
The conversion of 2-methylpropene to
To get the hydrogenated product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H−H bond of the reagent.
- Add one H atom to double-bonded C atom to form two new C−H single bonds in the molecule.
Hence the hydrogenation of 2-methylpropene can be written as:
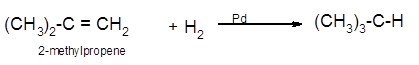
(c)
Interpretation:
The reagent that is needed to convert 2-methylpropene to
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
Hydrohalogenation reaction is an addition reaction in which the hydrogen and halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Answer to Problem 13.67P
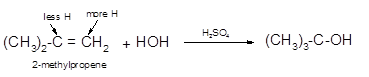
Explanation of Solution
The conversion of 2-methylpropene to
To get the hydration product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H-OH bond of the reagent.
- Add the -OH group atom to double-bonded C atom to form new C−OH single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-OH will bond to that double-bonded C atom which has more number of H atoms.
Hence, the hydration of 2-methylpropene can be written as:
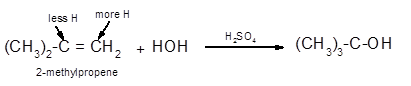
(d)
Interpretation:
The reagent that is needed to convert 2-methylpropene to
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
Hydrohalogenation reaction is an addition reaction in which the hydrogen and halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Answer to Problem 13.67P
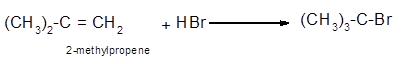
Explanation of Solution
The conversion of 2-methylpropene to
To get the hydrohalogenation product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H-Br bond of the reagent.
- Add one Br atom to double-bonded C atom to form new C−Cl single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-X will bond to that double-bonded C atom which has more number of H atoms.
Hence, the hydrohalogenation of 2-methylpropene can be written as:
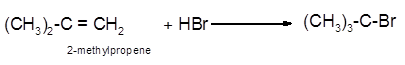
(e)
Interpretation:
The reagent that is needed to convert 2-methylpropene to
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
Hydrohalogenation reaction is an addition reaction in which the hydrogen and halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Answer to Problem 13.67P
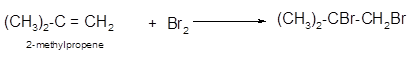
Explanation of Solution
The conversion of 2-methylpropene to
To get the halogenation product of any alkene, three steps must be followed:
- Locate the position of C=C in the molecule.
- Break the Br-Br bond of the reagent.
- Add one Br atom to double-bonded C atom to form two new C−Br single bonds in the molecule.
Hence, the halogenation of 2-methylpropene can be written as:
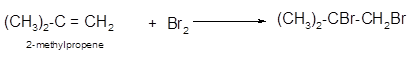
(f)
Interpretation:
The reagent that is needed to convert 2-methylpropene to
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
Hydrohalogenation reaction is an addition reaction in which the hydrogen and halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Answer to Problem 13.67P
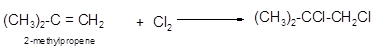
Explanation of Solution
The conversion of 2-methylpropene to
To get the halogenation product of any alkene, three steps must be followed:
- Locate the position of C=C in the molecule.
- Break the Cl-Cl bond of the reagent.
- Add one Cl atom to double-bonded C atom to form two new C−Cl single bonds in the molecule.
Hence the halogenation of 2-methylpropene can be written as follows:
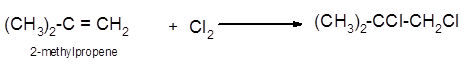
Want to see more full solutions like this?
Chapter 13 Solutions
ALEKS 360 ACCESS CARD F/GEN. ORG.CHEM
- 8. Give the IUPAC name for each alkene. CH3 CH2CH,CH3 Br а.arrow_forwarda. What hydrocarbon with molecular formula C4H10 forms only two monochlorinated products? Both products are achiral. b. What hydrocarbon with the same molecular formula as in part a forms three monochlorinated products? One is achiral and two are chiral.arrow_forward3. What alcohol is formed when each alkene is treated with H2O in the presence of H2SO4 (as the catalyst)? а. СНЗСН-СНСНЗ b. CH3CH2CH=CH2 С. CH3 CH3arrow_forward
- taken in order to gor the product Please explain the mechanisms/stepsarrow_forwardCH3 KOC(CH 3)3 H;C- -CH3 Solvent: Ethanol HCI c-OH Solvent: Ethanol CH3 KOC(CH 3)3 Solvent: Toluene H3C- -CH3 HCI CH3 но CH3 Solvent: Toluene H3C H3C CH3 KOC(CH 3)3 .CI Solvent: Toluene -CH3 CH3arrow_forward1. CH, CH, - CH,-CH,-CH, + HO 2. H₂C-CH=CH₂ + HBr/H₂O₂ 3. H₂C-CH (OH)-CH₂ + H* 4. Nitrobenzene +? ? anilinearrow_forward
- 6. What is the relationship between A and B? HO OH II A) They are constitutional isomers C) They are tautomers B) D) They are stereoisomers They are enantiomersarrow_forwardA. GIVE THE IUPAC NAME OF THE FR ОН b) (CH3)3СОН с) СНЗСН2СH2ОН d) OH .C. H3C CH3 H.arrow_forwardHCI CH3 но. CH3 Solvent: Toluene H;C H3C CH3 KOC(CH 3)3 .CI Solvent: Toluene -CH3 CH3arrow_forward
- Classify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forward'N' 80 Draw Enamine 1. CH3CH=CHC( O)CH3, heat 2. H3O+ Drawingarrow_forward1. Give IUPC name for the following compounds a) (CH3)3COH c) HOCH₂CH₂OH b) CH₂=CH-CH₂OH d) CH3CH(CH3)-CH₂OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





