
Concept explainers
(a)
Interpretation:
The product formed should be determined on the reaction of following

Concept Introduction:
A
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydration reaction is an addition reaction in which the hydrogen and hydroxyl group (-OH) are bonded on un-statured carbon atoms of alkene to form alcohol.
Answer to Problem 13.62P

Explanation of Solution
To get the hydration product of any alkene, three steps must be followed:
- Locate the position of C=C in the molecule.
- Break the H-OH bond of the reagent.
- Add the -OH group atom to double-bonded C atom to form new C−OH single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-OH will bond to that double-bonded C atom which has more number of H atoms.
Hence, the hydration product is:
. 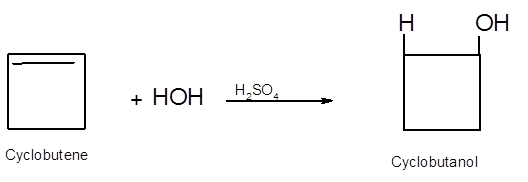
(b)
Interpretation:
The product formed should be determined on the reaction of following alkene with
(CH3)3 -C =C -(CH3)2
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydration reaction is an addition reaction in which the hydrogen and hydroxyl group (-OH) are bonded on un-statured carbon atoms of alkene to form alcohol.
Answer to Problem 13.62P
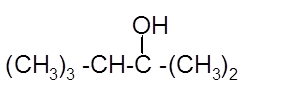
Explanation of Solution
To get the hydration product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H-OH bond of the reagent.
- Add the -OH group atom to double-bonded C atom to form new C−OH single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-OH will bond to that double-bonded C atom which has more number of H atoms.
Hence, the hydration product is:
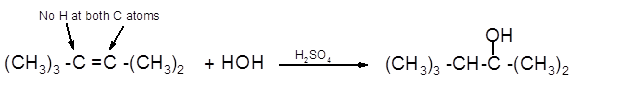
(c)
Interpretation:
The product formed should be determined on the reaction of following alkene with
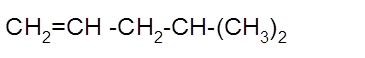
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydration reaction is an addition reaction in which the hydrogen and hydroxyl group (-OH) are bonded on un-statured carbon atoms of alkene to form alcohol.
Answer to Problem 13.62P
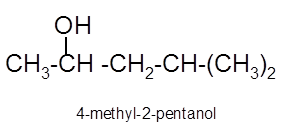
Explanation of Solution
To get the hydration product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H-OH bond of the reagent.
- Add the -OH group atom to double-bonded C atom to form new C−OH single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-OH will bond to that double-bonded C atom which has more number of H atoms.
Hence, the hydration product is:
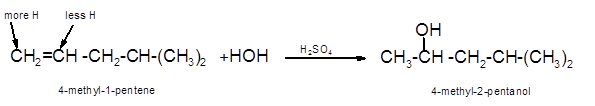
(d)
Interpretation:
The product formed should be determined on the reaction of following alkene with
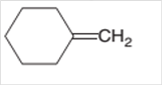
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant, whereas, the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydration reaction is an addition reaction in which the hydrogen and hydroxyl group (-OH) are bonded on un-statured carbon atoms of alkene to form alcohol.
Answer to Problem 13.62P
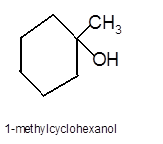
Explanation of Solution
To get the hydration product of any alkene, three steps must be followed;
- Locate the position of C=C in the molecule.
- Break the H-OH bond of the reagent.
- Add the -OH group atom to double-bonded C atom to form new C−OH single bonds in the molecule.
- Add one H atom to another double-bonded C atom to form new C−H single bonds in the molecule.
- The reaction follows the Markovnikov rule which states that the H atom of H-OH will bond to that double-bonded C atom which has more number of H atoms.
Hence the hydration product is:
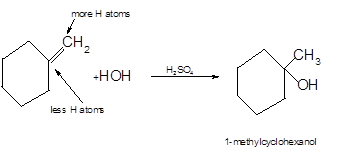
Want to see more full solutions like this?
Chapter 13 Solutions
ALEKS 360 ACCESS CARD F/GEN. ORG.CHEM
- What alkenes are formed when each alcohol is treated with H 2SO 4? Use the Zaitsev rule to predict the major product.arrow_forwardWhat alcohol is formed when the compound is treated with H2 and a Pd catalyst?arrow_forwardWhat alcohol is formed when the following compound is treated with H2 and a Pd catalyst?arrow_forward
- Give the IUPAC name of the alcohol that fits each of the following descriptions. a. Moistening agent in many cosmetics b. Major ingredient in environmentally friendly antifreeze formulations c. Industrially produced from CO and H2 d. Often produced via a fermentation processarrow_forward14-60 Write a balanced equation for the complete combustion of ethanol, the alcohol blended with gasoline to produce E85.arrow_forward14-39 Name two important alcohols derived from ethylene and give two important uses of each.arrow_forward
- In which solvent is cyclohexane most soluble? Water Diethyl ether Hexanol ethanolarrow_forwardWhat product is formed when each alkene is treated with H 2 and a Pd catalyst?arrow_forward3. Complete the following intramolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cy-C-CH-oH 18UC b) CHy CH-Cy-CHy 180C OH c) 180c 4. Complete the following intermolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy-CH-OH b) Ho SわふわSわ 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. a) hparrow_forward
- What alcohol is formed when each compound is treated with NaBH4 in CH3OH?arrow_forwardWhy can formaldehyde (CH20) be prepared in the form of a 37% solution in water, whereas octanal cannot? (Select all that apply.) | Octanal is more hydrophilic than formaldehyde. O Formaldehyde is a small molecule. O Formaldehyde is polar. O Octanal is mostly hydrophobic.arrow_forwardWhat products are formed when each alcohol is oxidized with K 2Cr 2O 7? In some cases, no reaction occurs.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


