
Concept explainers
(a)
Interpretation:
The complete reaction between
Concept introduction:
A
Answer to Problem 13.52E
The completed reaction between
Explanation of Solution
The incomplete reaction is shown below.
The thiols react with heavy metal ions such as
Therefore, the completed reaction between
The completed reaction between
(b)
Interpretation:
The complete reaction between
Concept introduction:
A chemical reaction is a process in which rearrangement of atoms or ions takes place between two reacting species. A balanced chemical equation represents an equation in which all the reactants and products are written with their stoichiometric coefficients. The number of atoms of an element on both sides of the equation is equal.
Answer to Problem 13.52E
The completed reaction between
Explanation of Solution
The incomplete reaction is shown below.
Two molecules of thiol undergo an oxidation reaction with a nascent oxygen atom. The thiol molecules react with nascent oxygen atom to form a disulfide compound. Therefore, the completed reaction between
The completed reaction between
(c)
Interpretation:
The complete reaction between given disulfide and nascent hydrogen atoms is to be stated.
Concept introduction:
A chemical reaction is a process in which rearrangement of atoms or ions takes place between two reacting species. A balanced chemical equation represents an equation in which all the reactants and products are written with their stoichiometric coefficients. The number of atoms of an element on both sides of the equation is equal.
Answer to Problem 13.52E
The complete reaction between given disulfide and nascent hydrogen atoms is shown below.

Explanation of Solution
The incomplete reaction is shown below.
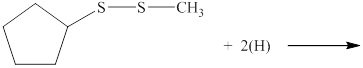
Figure 1
A disulfide compound undergoes reduction reaction with nascent hydrogen atoms to form two thiol molecules.
Therefore, the complete reaction between given disulfide and nascent hydrogen atom is shown below.

Figure 2
The complete reaction between given disulfide and nascent hydrogen atom is shown in Figure 2.
Want to see more full solutions like this?
Chapter 13 Solutions
Study Guide with Student Solutions Manual for Seager/Slabaugh/Hansen's Chemistry for Today: General, Organic, and Biochemistry, 9th Edition
- Complete the reaction by providing the starting material /reagent/products. Choose from the choices.arrow_forward2) Please complete the following syntheses A) Propene to 2-methyl-pent-2-ene B) pentan-2-amine to 3-bromopentanearrow_forwardGive the general formula for a normal alkene. OCH OCHAN OCH2 OCH 2 OCH 1arrow_forward
- Describe what is meant by a cis-isomer of an alkene.arrow_forward2. Draw out the condensed structural formulas for the following reactions. Name the organic product formed in each reaction. a) 3-methylpentanoic acid + KOH b) benzoic acid + 2-propanol (isopropyl alcohol) c) methanoic acid (formic acid) + 2-butanol (sec-butyl alcohol)arrow_forwardIn an esterification reaction, a carboxylic acid reacts with an excess of alcohol in acidic conditions to form an ester. Draw the structure of the ester product in the reaction between pentanoic acid and 1‑propanol.arrow_forward
- Complete the following organic reactions and draw structural diagrams to represent the products. a) CH3CH(CH3)CH2COOCH3 + H2O → b) methyl propene + HCl(aq) →arrow_forwardGive iupac name for this diol CH3CH(OH)(CH2)4CH(OH)C(CH3)3arrow_forwardb) Ho わ-fャコーfつ Koc 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. CHっーCHューC OH b) CHy CH3arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




