
Concept explainers
How would each of the following pairs of compounds differ in their IR spectra?
a. 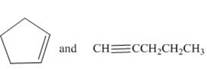 d.
d.![]()
b.![]() e.
e.
c.![]() f.
f.
(a)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR spectrum of cyclopentene is different from pentyne because the absorption of
Explanation of Solution
The given compounds are cyclopentene and pentyne as shown below.
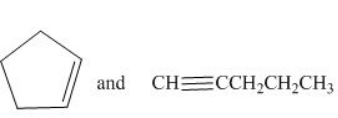
Figure 1
Cyclopentene contains
The IR spectrum of cyclopentene is different from pentyne because the absorption of
(b)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR absorption of propanoic acid is different from methyl acetate due to broad peak of
Explanation of Solution
The given compounds are propanoic acid and methyl acetate as shown below.
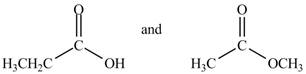
Figure 2
Propanoic acid contains
The IR absorptions of propanoic acid is different from methyl acetate due to broad peak of
(c)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR absorptions of butan-
Explanation of Solution
The given compounds are butan-

Figure 3
Butan-
The IR absorptions of propanoic acid is different from methyl acetate due to broad peak of
(d)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR spectra of methyl hexanoate are different from
Explanation of Solution
The given compound are
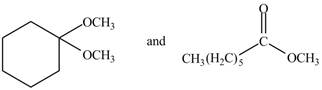
Figure 4
Methyl hexanoate contains
The IR spectra of methylhexanoate is different from
(e)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
But-
Explanation of Solution
But-
But-
(f)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR spectra of N, N-diethylprop-
Explanation of Solution
The given compound are N, N-diethylprop-
The IR spectra of N, N-diethylprop-
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry-Package(Custom)
- 8. Which molecule generated the following DEPT135 spectrum? CH3 CH3 CH3 OH H3C CH3 H3C CH3 H3CCH3 50 70 65 60 55 40 30 25 20 15 ppmarrow_forwardWhich of the indicated carbon atoms in each molecule absorbs farther downfield? a. CH,CH2OCH,CH3 b. BRCH,CHBr2 d. CH,CH=CH2 C. f OCH,arrow_forward7. How could you distinguish between 1-butanol and diethyl ether by infrared spectroscopy? 1-butanol OH both C4H10O 8. How could you distinguish between methyl butanoate and pentanoic acid by infrared spectroscopy? ha pentancic acid diethyl ether OH both CsH100 OCH3 methyl butanoatearrow_forward
- Which of the following molecules produced the 1H NMR spectrum shown below? (key: s = singlet, d = doublet, t = triplet, q = quartet, sept = septet, dd = doublet of doublets, br = broad singlet, m = multiplet) 2H, q 1H, q Cil 1H, br PPM 8 A. B. C. D. 2H, d 2H, d MeO 7 6 OH OH 3H, d 2 3H, tarrow_forward22. Which of the following compounds gives an infrared Spectrum with peaks at 3300cm (sharp Peak) and 2150 cm (sharp Peak) ? H LH ₂ CH ₂ C = CH CH3C=CCH 3 2 1 HCCH H₂C A) I B)2 c) 3 04 CAZ H 9 CH₂arrow_forward71. Which of (a)-(d) indicates the correct order of carbon chemical shifts of the four carbons of the following compound. II CH2=CH-C-0-CH3 C3 C2 C1 Сме А. СМе < С2 < С3 <С1 В. СМе <С3 < С2 < C1 C. CMe < C2 < C1 < C3 D. СМearrow_forwardQ4) Two Isomers have the chemical formula C,H, identify their structure based on their IR spectra below? Isomer A Ahserbane/ 3000 200 W es/e Isomer B Aboerkance/4arrow_forward5) Which one of the given compounds has a structure consistent with the IR spectrum shown? Clearly circle your choice. H₂N 100 TRANSMETTENCES D 4000 3000 H₂N 2 2000 AVENUMBER IN HN. 1500 H N H3C 1000 500 HO NHarrow_forwardWhat is the structure from the formula C10H12 and the spectra?arrow_forwardThe DEPT-90 spectrum exhibits 6 in the 0-50 ppm region The DEPT-135 spectrum exhibits x 100 ppm region that is a positive ▾ C6 signal(s) for the CH groups: ▼ 1,2,6 ✓ in the sp2 hybridized region 100-150 C3 and C4 ▼ signal(s) (only the quaternary carbon atoms, signal(s), indicating the presence of a methylene group (CH₂) attached to an oxygen atom, are missing); there is C5 ▼ C1 and C2 ▼ and signal(s) in the 50-arrow_forwardCH3CH2 -OH CH3- OCH3 A B -OCH2CH2CH3 C Which compound, if any, can be distinguished from the others by the molecular ions in their mass spectra? OB OA O None, they all have the same molar mass. осarrow_forwardational nown b elow. Vibrations of XEC14: Left: Scissoring. Middle: Wagging. Right: Twisting. Which of these motions will be IR active? In other words, which of these motions will lead to the absorption of IR light and be represented by a peak in the IR spectrum of this compound? O A. Only twisting is IR active. O B. Only wagging is IR active O C. Both scissoring and twisting are IR active. D. Both wagging and twisting are IR active. O E. All of these vibrations are IR activearrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
