
Concept explainers
Propose a structure consistent with each set of data.
a. a compound that contains a benzene ring and has a molecular ion at
b. a hydrocarbon that contains only
c. a compound that contains a carbonyl group and gives a molecular ion at
d. a compound that contains
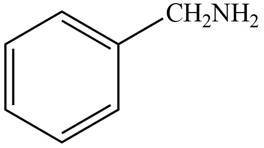
(a)
Interpretation: The structure of a compound that contains a benzene ring and has a molecular ion at
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.33P
The structure of the compound that contains a benzene ring and has a molecular ion at
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
Thus, the possible molecular formula is
The molecular ion has odd mass. Thus, the compound may contain
Possible compounds with
• Substitute
Thus, the possible molecular formula is
The molecular mass of
Hence, the structure of a compound that contains a benzene ring and has a molecular ion at
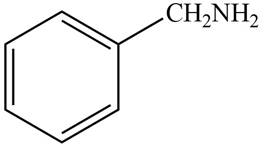
Figure 1
The structure of a compound
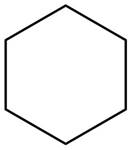
(b)
Interpretation: The structure of a hydrocarbon that contains only
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.33P
The structure of a hydrocarbon
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
• Replace one carbon by
Thus, the possible molecular formula is
The molecular mass of
Hence, the structure of a hydrocarbon that contains only
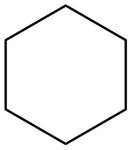
Figure 2
The structure of a hydrocarbon
(c)
Interpretation: The structure of a compound that contains a carbonyl group and gives a molecular ion at
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.33P
The structure of a compound
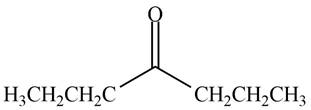
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
• Replace one carbon by
The compound that contain
Possible compounds with
• Substitute
Thus, the possible molecular formula is
Hence, the structure of a compound that contains a carbonyl group and gives a molecular ion at
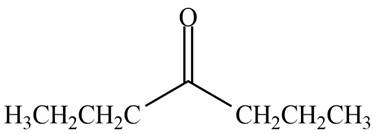
Figure 3
The structure of a compound
(d)
Interpretation: The structure of a compound that contains
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.33P
The structure of a compound
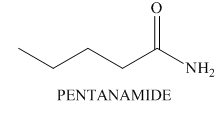
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
• Replace one carbon by
The given compound contains
Possible compounds with
• Substitute
• Substitute
Thus, the possible molecular formula is
Hence, the structure of a compound that contains

Figure 4
The structure of a compound
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry-Package(Custom)
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
Chemistry
General, Organic, & Biological Chemistry
- Determine the molecular formula for each of the following: a. a compound that contains only C and H and has a molecular ion with an m/z value of 72 b. a compound that contains C, H, and one O and has a molecular ion with an m/z value of 100 c. a compound that contains C, H, and two Os and has a molecular ion with an m/z value of 102 d. an amide that has a molecular ion with an m/z value of 115arrow_forwardAcetyleugenol: Match the peaks to the appropriate number on the structure. A letter may correspond to more than one number. 10 11 2. 2 3. 3 44 5. 5 6. 6arrow_forwardMatch each spectrum to the isomers of xylene: ortho-xylene, meta-xylene and para-xylene. (ignore the CDCI3 signal) Spectrum A CH3 CH 3 o-xylene 200 Spectrum B 200 spectrum C CH3 CH3 m-xylene 100 100 CH 3 CH 3 p-xylenearrow_forward
- Which of the indicated carbon atoms in each molecule absorbs farther downfield? a. CH,CH2OCH,CH3 b. BRCH,CHBr2 d. CH,CH=CH2 C. f OCH,arrow_forwardFigure 1.9: The structure of this acid (C5H10O2) is: 4 singlet triplet 3 doublet 3 n=8 Figure 3: ¹H-NMR of unknown ether C (molecular formula C5H12O). Figure 1.10: The structure of this ether (C5H10O2) is: Figure 1.11: The structure of this compound (C7H16) is: мий doublet n=8 ми ми doublet triplet 1arrow_forwardSelect the best pair of answer below justifying the reason for your choice. Answer Q48 & 49arrow_forward
- Which atom do you expect is present in this spectrum? 100 Rel. Intensity 80 60 60 40 40 20 20 0.0 0.0 15 30 45 60 75 90 m/z NIST Chemistry WebBook (https://webbook.nist.gov/chemistry) Only hydrocarbons Bromine Chlorine O Nitrogenarrow_forwardAccording to the conventions above, what is the sign ( + or ) of the P.E. change (H) for Rxn 3?arrow_forward8. For the mass spec shown below, select the structure that matches. How can you tell? MASS SPECTRUM mie RA 72 15 73 12 74 010 50 70 30 90 10 m/e CH2 CH2 CH2 CH3 CH2 H3C° CH2 NH2 H. CH3 NH2 CH3 CH3 CH2 CH H3C CH2 H3Carrow_forward
- Isoamyl acetate: Match the peaks to the appropriate number on the structure. A letter may correspond to more than one number. e 5 9 a b c d d. I 3. 6. 200 180 160 140 120 100 80 60 40 4 g 1. 1 3_ a 2. 2 5 e 3. 3 5 f 4. 4 1 b 2 c 5. 5 6. 6 6 d 20arrow_forwardWhich structure is consistent with an [M] peak and an [M+2]" peak in the ratlo of 3:17 Click on a letter A through C to answer, А. Br CI В. NH2 С.arrow_forwardQUESTION 1 consistent with Which of the following spectroscopic measurements cyclohexanecarbaldehyde? is not O A. H-NMR: a doublet (J = 1.3 Hz) at 9.6 ppm O B. MS: a parent peak at m/z 98. O C. C-NMR: a peak at 204.7 ppm that has 1 H attached OD. IR: peaks at 2856 cm1 (6% transmittance); 2809 cm1 (37%); 2705 cm1 (35%), and 1727 cm-1 (5%).arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

