
(a)
Interpretation:
The given statements have to be answered.
Concept Introduction:
The time taken by the concentration of reaction to get reduced of its original concentration is called as half-life reaction.
Half life for first order reactions:
The half life for the first order reaction is constant and it is independent of the reactant concentration.
Half life period of first order reaction can be calculated using the equation,
Half life for second order reactions:
In second order reaction, the half-life is inversely proportional to the initial concentration of the reactant (A).
The half-life of second order reaction can be calculated using the equation,
Since the reactant will be consumed in lesser amount of time, these reactions will have shorter half-life.
To complete the pictures
(a)
Explanation of Solution
The reaction follows first order with presence of half-life of ten seconds.
There are 16 AB particles present in the container,
After one half life (10s) 8 particles will be reacted and 8 remains unreacted.
After two-half lives (20s) 12 particles will be reacted and 4 remains unreacted.
The completed pictures are,
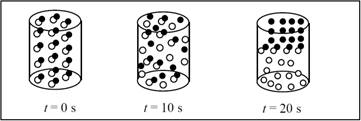
Figure 1
(b)
Interpretation:
The given statements have to be answered.
Concept Introduction:
The time taken by the concentration of reaction to get reduced of its original concentration is called as half-life reaction.
Half life for first order reactions:
The half life for the first order reaction is constant and it is independent of the reactant concentration.
Half life period of first order reaction can be calculated using the equation,
Half life for second order reactions:
In second order reaction, the half-life is inversely proportional to the initial concentration of the reactant (A).
The half-life of second order reaction can be calculated using the equation,
Since the reactant will be consumed in lesser amount of time, these reactions will have shorter half-life.
To explain the changes in completed figure if the reaction was second-order with same half life
(b)
Explanation of Solution
If the half-life is similar for second-order reaction, the container t=20s would have more number of AB and fewer A and B when compared to part a.
(c)
Interpretation:
The given statements have to be answered.
Concept Introduction:
The time taken by the concentration of reaction to get reduced of its original concentration is called as half-life reaction.
Half life for first order reactions:
The half life for the first order reaction is constant and it is independent of the reactant concentration.
Half life period of first order reaction can be calculated using the equation,
Half life for second order reactions:
In second order reaction, the half-life is inversely proportional to the initial concentration of the reactant (A).
The half-life of second order reaction can be calculated using the equation,
Since the reactant will be consumed in lesser amount of time, these reactions will have shorter half-life.
To give the relative
(c)
Explanation of Solution
After 10 seconds, the concentration of the particles is one-half their initial value. Then relative rate of reactions for first-order at the start and after 10 seconds are,
(d)
Interpretation:
The given statements have to be answered.
Concept Introduction:
The time taken by the concentration of reaction to get reduced of its original concentration is called as half-life reaction.
Half life for first order reactions:
The half life for the first order reaction is constant and it is independent of the reactant concentration.
Half life period of first order reaction can be calculated using the equation,
Half life for second order reactions:
In second order reaction, the half-life is inversely proportional to the initial concentration of the reactant (A).
The half-life of second order reaction can be calculated using the equation,
Since the reactant will be consumed in lesser amount of time, these reactions will have shorter half-life.
To give the relative reaction rates for second order reaction at the start of reaction and after 10 seconds elapsed
(d)
Explanation of Solution
After 10 seconds, the concentration of the particles is one-half their initial value. Then relative rate of reactions for second order at the start and after 10 seconds are,
Want to see more full solutions like this?
Chapter 13 Solutions
General Chemistry
- Derive an expression for the half-life of a a third order reaction;b a reaction whose order is =1; c a reaction whose order is 12. In these last two cases, examples are rare but known.arrow_forwardExplain why half-lives are not normally used to describe reactions other than first order.arrow_forwardComplete the following table for the reaction below. It is first-order in both X and Y. 2X(g)+Y(g)productsarrow_forward
- Make a graph of [A] versus time for zero-, first-, and second-order reactions. From these graphs, compare successive half-lives.arrow_forwardConsider the hypothetical first-order reaction 2A(g)X(g)+12Y(g)At a certain temperature, the half-life of the reaction is 19.0 min. A 1.00-L flask contains A with a partial pressure of 622 mm Hg. If the temperature is kept constant, what are the partial pressures of A, X, and Y after 42 minutes?arrow_forwardThe hydrolysis of the sugar sucrose to the sugars glucose and fructose, C12H22O11+H2OC6H12O6+C6H12O6 follows a first-order rate equation for the disappearance of sucrose: Rate =k[C12H22O11] (The products of the reaction, glucose and fructose, have the same molecular formulas but differ in the arrangement of the atoms in their molecules.) (a) In neutral solution, k=2.11011s1 at 27 C and 8.51011s1 at 37 C. Determine the activation energy, the frequency factor, and the rate constant for this equation at 47 C (assuming the kinetics remain consistent with the Arrhenius equation at this temperature). (b) When a solution of sucrose with an initial concentration of 0.150 M reaches equilibrium, the concentration of sucrose is 1.65107M . How long will it take the solution to reach equilibrium at 27 C in the absence of a catalyst? Because the concentration of sucrose at equilibrium is so low, assume that the reaction is irreversible. (c) Why does assuming that the reaction is irreversible simplify the calculation in pan (b)?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





