
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.5, Problem 8P
What products are formed when the following compounds react with CH3MgBr, followed by the addition of dilute acid? Disregard stereoisomers.
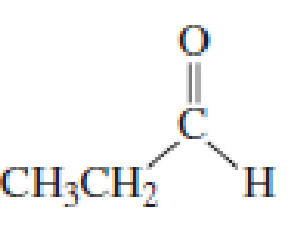
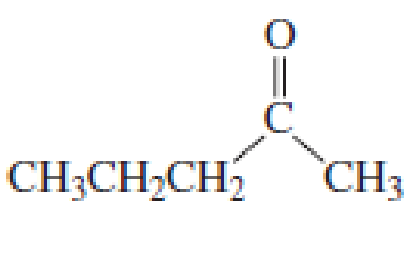
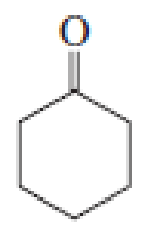
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Two products are formed when methylenecyclohexane reacts with NBS? Show how each is formed. Disregard stereoisomers.
What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at room temperature? Disregard stereoisomers.
How many stereoisomers are obtained from the reaction of 3-methyl-2-butanone with each of the following organometallic reagents followed by the addition of dilute acid? Name each stereoisomer.
ethyllithium
methylmagnesium bromide
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 12.1 - Prob. 1PCh. 12.1 - Name the following compounds:Ch. 12.1 - Prob. 3PCh. 12.1 - Prob. 4PCh. 12.2 - Prob. 5PCh. 12.4 - Draw the structure for each of the following: a....Ch. 12.4 - Prob. 7PCh. 12.5 - What products are formed when the following...Ch. 12.5 - Prob. 9PCh. 12.5 - Prob. 10P
Ch. 12.5 - Prob. 12PCh. 12.5 - Write the mechanism for the reaction of acetyl...Ch. 12.5 - Prob. 14PCh. 12.6 - Prob. 16PCh. 12.7 - Prob. 17PCh. 12.7 - Prob. 18PCh. 12.7 - Prob. 19PCh. 12.8 - Prob. 20PCh. 12.8 - Prob. 21PCh. 12.8 - Prob. 22PCh. 12.9 - Which of the following are a. hemiacetals? b....Ch. 12.9 - Prob. 24PCh. 12.9 - Prob. 25PCh. 12.10 - Prob. 26PCh. 12.11 - Prob. 27PCh. 12 - Draw the structure for each of the following: a....Ch. 12 - Prob. 29PCh. 12 - List the following compounds in order from most...Ch. 12 - Show the reagents required to form the primary...Ch. 12 - Fill in the boxes:Ch. 12 - Indicate how the following compounds could be...Ch. 12 - Prob. 34PCh. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - Prob. 37PCh. 12 - Prob. 38PCh. 12 - Prob. 39PCh. 12 - Show two ways the following compound could be...Ch. 12 - Prob. 41PCh. 12 - Prob. 42PCh. 12 - Prob. 43PCh. 12 - Prob. 44PCh. 12 - Prob. 45PCh. 12 - Prob. 46PCh. 12 - Prob. 47PCh. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - Prob. 50PCh. 12 - Prob. 51PCh. 12 - Indicate how the following compounds could be...Ch. 12 - Prob. 53P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Cyclohexene can be converted to 1-cyclopentenecarbaldehyde by the following series of reactions. Propose a structural formula for each intermediate compound.arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardFollowing is the structural formula of Surfynol, a defoaming surfactant. Describe the synthesis of this compound from acetylene and a ketone. How many stereoisomers are possible for Surfynol?arrow_forward
- Cyclohexanone reacts with NaCN to form The S-isomer The R-isomer A racemic mixture None of the abovearrow_forwardWhat is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at roomtemperature? Disregard stereoisomers.arrow_forwardWhat is the major product formed when 3-methyl-1-butene is treated with Hg(C2H3O2)2 in a THF/ethanol mixture followed by NaBH4 in base?arrow_forward
- Predict the products formed when cyclohexanone reacts with the following reagents.(a) CH3NH2, Harrow_forwardGive the structures of the substitution products expected when 1-bromohexane reacts with(a) NaOCH2CH3 (b) KCN (c) NaOHarrow_forwardPredict the major products of the following reactions, including stereochemistry where appropriate. (a) n-butanol + HBr (b) cyclooctylmethanol + CH3CH2MgBrarrow_forward
- Name and draw the structure of the major product in the following reactions of cyclohexanone: A) Reduction with NaBH4 in ethanol, followed by H+ B) Reaction with excess ethanol in the presence of H+arrow_forwardWhich one(s) of the following reactions produce propan-1-ol? A) the alkaline hydrolysis of 1-chloropropane B) the acid hydrolysis of propyl methanoate C) the acid hydrolysis of propanenitrilearrow_forwardStarting with cyclohexanone, show how to prepare these compounds. In addition to the given starting material, use any other organic or inorganic reagents as necessary. Q.) 1-Methylcyclohexenearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY