
Concept explainers
(a)
Interpretation:
To identify the products are formed when 3-methyl-2-cyclohexenone reacts with each of the given reactant.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as
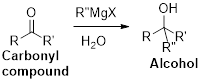
(b)
Interpretation:
To identify the products are formed when 3-methyl-2-cyclohexenone reacts with each of the given reactant.
Concept introduction:
Michael reaction:
The nucleophile is reaction with α,β-unsaturated carbonyl compound (1,4 –addtion) which yield the addition product is called Michael reaction.
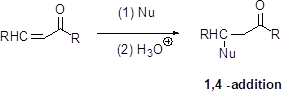
(c)
Interpretation:
To identify the products are formed when 3-methyl-2-cyclohexenone reacts with each of the given reactant.
Concept introduction:
Michael reaction:
The nucleophile is reaction with α,β-unsaturated carbonyl compound (1,4 –addtion) which yield the addition product is called Michael reaction.
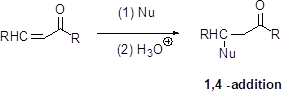
Anti-Markovnikov’s rule: unsymmetrical
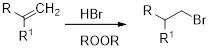
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
- Consider the structure of pent-2-ene, if it undergoes ozonolysis, which of the following final product is formed? a.Ethanal and Propanal b.Ethanal and Propanone c.CO2 and Propanal d.Ethanal and CO2arrow_forwardDraw the product formed when pentanal (CH3 CH₂ CH₂ CH₂ CHO) is treated with each reagent. With some reagents, no reaction occurs. a. NaBH4, CH3OH b. [1] LiAiH4: [2] H₂O c. H₂, Pd-C d. PCC e. Na₂Cr₂O7, H₂SO4, H₂O f. Ag₂O, NH4OH g. [1] CH3 MgBr; [2] H₂O h. [1] C6H5 Li: [2] H₂O i. [1] (CH3)2 CuLi; [2] H₂O J. [1] HC=CNa; [2] H₂O k. [1] CH 3 C=CLI; [2] H₂O 1. The product in (a), then TBDMS-CI, imidazolearrow_forwardWhat alkenes are formed when each alcohol is dehydrated with TSOH? Label the major product when a mixture results. OH Xom OH a. OH b. CH₂CH3 OH C. d. CH₂CH₂CH₂CH₂OH e.arrow_forward
- Draw out each compound to clearly show what groups are bonded to the carbonyl carbon. Label each compound as a ketone or aldehyde. a. CH 3CH 2CHO b. CH 3CH 2COCH 3 c. (CH 3) 3CCOCH 3 d. (CH 3CH 2) 2CHCHOarrow_forwardWhat ketone could be used to prepare 3-methylpentan-3-ol via a Grignard reaction with CH3MgBr? a.) Pentan-2-oneb.) Pentan-3-onec.) Pentan-4-one d.) acetonearrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forward
- 10. Write chemical equations for the reaction of ethyl propanoate with each reagent below. a) LIAIH4/ H3O* b) NH3 d) NaOH c) C2H5NH2arrow_forwardDraw the major organic product for each of the following reactions. 1. ВНз. THF 2. NaOH, H-O, НаО2 1. Hg(OAc)2, H20 2. NaBH4 H2SO4 H20arrow_forwardDraw the products formed when phenol (C6H5OH) is treated with each set of reagents. a. [1] HNO3, H2SO4; [2] Sn, HCl b. [1] (CH3CH2)2CHCOCl, AlCl3; [2] Zn(Hg), HCl c. [1] CH3CH2Cl, AlCl3; [2] Br2, hν d. [1] (CH3)2CHCl, AlCl3; [2] KMnO4arrow_forward
- Provide the major organic product of the following reaction. but-1-yne 1) Sia₂BH 2) H₂O₂, OH What is it called when an enol converts to a keto? A) Keto-enol isomerism B) Keto-enol tater tot-ism C) Keto-enol conversion D) Keto-enol tautomerismarrow_forwardDraw the products formed when each compound is treated with HNO3 and H2SO4.State whether the reaction occurs faster or slower than a similar reaction with benzene.arrow_forwardWhich carbonyl group is least reactive for nucleophilic addition or substitution reactions? H2N III H. IV O V only O Il only O IV only O I only O II onlyarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





