
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
13th Edition
ISBN: 9780134421353
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12.4, Problem 12.32PP
Draw the condensed structural formulas for the
a.
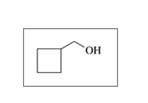
b. CH3-OH
c 3-chloro-1-propanol
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the structure of each of the following carboxylic acids:a. b-Chlorobutyric acidb. a,b-Dibromovaleric acidc. b,g-Dihydroxybutyric acidd. d-Bromo-g-chloro-b-methylcaproic acid
Draw the condensed structural formula of the organic product formed when each of the following is reduced by hydrogen in the presence of a nickel catalyst:a. ethyl propyl ketone
b. formaldehydec. 3-chlorocyclopentanoned. 2-pentanone
69. The structure of the aldehyde or ketone that the
following compound came from:
H
A. III only
B. IV only
C. I and IV
D. II only
IV
Chapter 12 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Prob. 12.3PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Give the common name for each of the following: a....Ch. 12.1 - Prob. 12.6PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Draw the condensed structural formula, or...Ch. 12.2 - Classify each of the following alcohols as primary...Ch. 12.2 - Classify each of the following alcohols as primary...
Ch. 12.2 - Prob. 12.11PPCh. 12.2 - Prob. 12.12PPCh. 12.2 - Give an explanation for each of the following...Ch. 12.2 - Give an explanation for each of the following...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Prob. 12.27PPCh. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Prob. 12.33PPCh. 12.4 - Prob. 12.34PPCh. 12.4 - Oxybenzone is an effective sunscreen whose...Ch. 12.4 - Avobenzone is a common ingredient in sunscreen....Ch. 12 - Prob. 12.37UTCCh. 12 - The compound frambinone has the taste of...Ch. 12 - A compound called resveratrol is an antioxidant,...Ch. 12 - A compound called cinnamaldehyde is found in...Ch. 12 - Prob. 12.41UTCCh. 12 - Prob. 12.42UTCCh. 12 - Prob. 12.43APPCh. 12 - Classify each of the following alcohols as primary...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Prob. 12.63CPCh. 12 - Draw the condensed structural formulas and give...Ch. 12 - A compound with the formula C4H8O is synthesized...Ch. 12 - A compound with the formula C5H10O oxidizes to...Ch. 12 - Compound A is a primary alcohol whose formula is...Ch. 12 - Compound X is a secondary alcohol whose formula is...Ch. 12 - Prob. 21CICh. 12 - Prob. 22CICh. 12 - Prob. 23CICh. 12 - Prob. 24CICh. 12 - Prob. 25CICh. 12 - lonone is a compound that gives violets their...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the complete chemical reaction and correct chemical formula and structure of the following: 1. Autooxidation of benzaldehyde 2. reaction between acetic acid and ethanol 3. reaction between aspirin and sodium hydroxide 4. reaction of acetone with methanolarrow_forwardDraw condensed formulas for each of the following compounds:a. Methyl benzoateb. Butyl decanoatec. Methyl propionated. Ethyl propionatearrow_forwardWrite an equation representing the neutralization of eachof the following carboxylic acids with KOH:a. 3-Chlorohexanoic acidb. Cyclohexanecarboxylic acidc. 3,4-Dimethylpentanoic acidarrow_forward
- Grignard reagents react with aldehyde to form: а. Carbon dioxide b. Secondary alcohol С. Ketone d. Carboxylic acidarrow_forwardCarboxylic Acid Draw the condensed formula of the following compounds: 7. nonanoic acid8. g-sec. butylcaprylic acid9. 4-ethyl-3-methylheptanedioic acidarrow_forwardWrite a balanced equation for the hydrogenation of eachof the following:a. Propanal (a three-carbon aldehyde)b. Propanone (a three-carbon ketone)c. 2,3-Dimethylheptanal (an aldehyde with a seven-carbon parent chain)d. 3-Methyl-4-heptanone (a ketone with a seven-carbonparent chain)arrow_forward
- I 2. Draw the structural formula of the organic product when each of the following aldehydes is oxidized to a carboxylic acid. Example: CH,-C-OH Ethanal ANSWER: a. Pentanal p. Formaldehyde . 3,4-Dichlorohexanalarrow_forwardChoose the correct IUPAC name for 2-Isopropyl-4-methylheptane. O A. 4,6,7-trimethyloctane O B. 4,5-dimethyl-2-propylhexane O C. 2,3,4-trimethyloctane O D. 2-isopropyl-4-methylheptanearrow_forwardDraw structural formulas for the following carboxylic acids and circle the acidic hydrogen atoms present: a. pentanoic acid b. 3,4-dichlorobutanoic acid c. 3,3,5-trimethylhexanoic acidarrow_forward
- 23 Classify the following compound: A. Ketal B. Acetal C. Hemiketal D. Hemiacetal CH 3 O Harrow_forwardDraw the condensed formulas for each of the following compounds: (a) dipropyl ether. (b) 2, 2-dimethyl-3-hexanol. (c) 2-ethoxybutanearrow_forwardDraw the condensed formula of the following compounds: 7. nonanoic acid8. g-sec. butylcaprylic acid9. 4-ethyl-3-methylheptanedioic acidarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY