
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
13th Edition
ISBN: 9780134421353
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.2, Problem 12.10PP
Classify each of the following alcohols as primary (1°), secondary (2°), or tertiary (3°):
a.
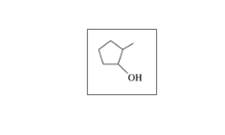
b.
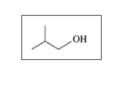
c.
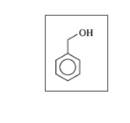
d.
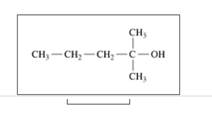
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. Name the following alcohols according to IUPAC rules:
a.
NO₂
b.
C.
OH
Name:
CI
OH
OH
Name:
Name:
OH
which include halogenated hydrocarbons.
10. The
alcohols, ethers, phenols. and amines.
O carbonyl functional groups
O non-carbonyl functional groups
O alkyl groups
11.
is largely used for the production of acetic acid and pyridines
derivatives.
Acetaldehyde
Benzaldehyde
Formaldehyde
Define the following terms: a. reaction mechanism b. carbocation c. enediol d. general acid e. general base
Chapter 12 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Give the IUPAC name for each of the following: a....Ch. 12.1 - Prob. 12.3PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Give the common name for each of the following: a....Ch. 12.1 - Prob. 12.6PPCh. 12.1 - Draw the condensed structural formula, or...Ch. 12.1 - Draw the condensed structural formula, or...Ch. 12.2 - Classify each of the following alcohols as primary...Ch. 12.2 - Classify each of the following alcohols as primary...
Ch. 12.2 - Prob. 12.11PPCh. 12.2 - Prob. 12.12PPCh. 12.2 - Give an explanation for each of the following...Ch. 12.2 - Give an explanation for each of the following...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Identify each of the following compounds as an...Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the common name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Give the IUPAC name for each of the following: a....Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.3 - Which compound in each of the following pairs...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Write the balanced chemical equation for the...Ch. 12.4 - Prob. 12.27PPCh. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural or line-angle...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Draw the condensed structural formulas for the...Ch. 12.4 - Prob. 12.33PPCh. 12.4 - Prob. 12.34PPCh. 12.4 - Oxybenzone is an effective sunscreen whose...Ch. 12.4 - Avobenzone is a common ingredient in sunscreen....Ch. 12 - Prob. 12.37UTCCh. 12 - The compound frambinone has the taste of...Ch. 12 - A compound called resveratrol is an antioxidant,...Ch. 12 - A compound called cinnamaldehyde is found in...Ch. 12 - Prob. 12.41UTCCh. 12 - Prob. 12.42UTCCh. 12 - Prob. 12.43APPCh. 12 - Classify each of the following alcohols as primary...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Give the IUPAC name for each of the following...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Which compound in each pair would be more soluble...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Which of the following aldehydes or ketones are...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Draw the condensed structural or line-angle...Ch. 12 - Prob. 12.63CPCh. 12 - Draw the condensed structural formulas and give...Ch. 12 - A compound with the formula C4H8O is synthesized...Ch. 12 - A compound with the formula C5H10O oxidizes to...Ch. 12 - Compound A is a primary alcohol whose formula is...Ch. 12 - Compound X is a secondary alcohol whose formula is...Ch. 12 - Prob. 21CICh. 12 - Prob. 22CICh. 12 - Prob. 23CICh. 12 - Prob. 24CICh. 12 - Prob. 25CICh. 12 - lonone is a compound that gives violets their...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Answer the following: 1. Which type of alcohol generally exhibits a higher dipole moment relative tolinear alcohol? a. Allylic alcoholb. Secondary alcoholc. Cyclic alkyl alcohold. Tertiary alcohol 2. What type of bond promotes the solubility of alcohols in water? a. Covalent bondsb. Van der Waals forcesc. Hydrogen bondsd. Dipole-dipole bonds 3. The delighted odor of melted butter is attributed to this four-carbon dione.What could be the IUPAC NAME of this ketone? a. Pentanedioneb. Hexanonec. Butanoned. Butanedione 4. Why is ethanal incapable of forming a hydrogen bond with another ethanalmolecule despite exhibiting a bond dipole in the C = O bond? a. The carbon atom does not form a highly polar bond with hydrogen.b. The oxygen atom is sp2 hybridized with almost 67% p character.c. The oxygen atom is partially reactive due to the strong bond dipole.d. Oxygen exhibits resonance with carbon leading to bond stability.arrow_forwardA sample consists of butyric acid, acetic acid and ethylene glycol. At a distilling temperature of 162°C, the distillate will significantly consist of: I. Butyric acid (boiling point: 162°C)II. Acetic acid (boiling point: 118°C)III. Ethylene glycol (boiling point: 197°C)A. I and II only B. I onlyC. I,II, and IIID. III onlyarrow_forwardINSTRUCTION: Give the IUPAC name for each ketone. A. B. C. D. E. CH₂CH₂CCHCH₂CH₂CH₂ SCHICHS CH₂ CH3 (CH₂)2CCCH₂CH₂CH₂CH3 CH₂ INSTRUCTION: DRAW the structure corresponding to each name. A. butyl ethyl ketone B. 2-methyl-3-pentanone C. p-ethylacetophenone D. 2-propylcyclobutanone E. 2-cyclohexenonearrow_forward
- 1. Naming 1. Provide the IUPAC name for each of the following compounds. a. b. Fu А 2. Provide the correct structure according to the following names. a. dibenzyl ether b. 1-methoxypentane c. diisopropyl etherarrow_forward10.38 Give the IUPAC name for each compound. a. b. C. d. e. OH f. OHarrow_forwardB. Describe each of the following: a. technical/commercial reagents b. U.S.P and N.F. reagents C. chemically pure reagents d. reagent grade chemicals e. primary standardsarrow_forward
- 1. An alkene reacts with water with an acid catalyst results into a formation of: A. Aldehyde B. Ketone C. Alcohol D. Ester 2. 3-Methylhexanal with K2Cr2O7 will yield: A. 3-Methyl-1-hexanol B. 3-Methylhexanoic acid C. 3-Methyl-1-hexanone D. 3-Methyl-1-hexanethiol 3. This is a reverse process of Hydration reaction: A. Oxidation reaction B. Reduction reaction C. Dehydration reaction D. Hydration reaction 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forward3. Draw structure s corresponding to the following IUPAC names: A. 2-methylhexan-2-ol B. Hexan-1,5-diol C. 2-ethylbut-2-en-1-ol D. Cyclohex-3-en-1-ol E. o-Bromophenol F. 2,4,6-trinitrophenolarrow_forwardDraw the following: 1. a. 3-methyl-1-butanethiol (skunk scent) b. triphenylmethanol c. 4-(bromomethyl)-3-octanol d. 3-cyclopentenethiolarrow_forward
- 1. Provide the IUPAC name for each of the following organic molecules. a. b. C. H3C H3C, Home H3C CH3 CH3 CH3 Br H 2. Draw structures for the following organic molecules, using the wedged/hashed line convention to depict any stereochemical relationships between groups: a. cis-3-ethylcyclobutane-1-carbaldehyde b. (R)-1-chloro-6-iodo-6-methyloctan-3-onearrow_forward1. For each reaction, write the letter corresponding to the type of reaction in column I and predict the MAJOR product. Draw the structure of the product in column III. A. Addition Reaction B. Electrophilic Aromatic Substitution Column I A. B. B. A. A. B. B. A. A. A. B. A. B. A. B. H₂C H3C H3C. H3C H3C H3C H3C H₂C CH3 CH3 H3C CH3 Reaction CH3 CH3 CH3 CH₂ ECH CH3 CH CH3 l CH3 CH₂ oso NH OEt CH3 CH3 orada CH3 CH3 -CH3 HNO3 H₂SO4 CH3 Br₂ DCM Br₂ hv H₂SO₂, H₂O HgSO4 Br₂ AlBr CH3 NBS hv HCI AICI Na₂Cr₂O7 H₂SO4, H₂O Br₂ FeBr H₂O* H₂ Lindlar's Catalyst 1) NaNH, 2) CH₂CH₂CH₂Br 3) H₂, Lindlar's catalyst CH3 NBS hv HNO, H₂SO4 C. Free Radical Substitution D. Redox Reaction Product (Column III)arrow_forwardInstructions: Give the IUPAC name for each compound. A. CH₂ CH₂CH₂CH₂CCH₂COOH CH₂ B. CH.CHCHCH.COOH a CH₂CH₂ C. (CH,CH,), CHCH,CH COOH Instructions: Give the structure corresponding to each IUPAC name. a. 2-bromobutanoic acid b. 2,3-dimethylpentanoic acid c. 2-ethyl-5,5-dimethyloctanoic acid d. 3,4,5,6-tetraethyldecanoic acidarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License