
(a)
Interpretation:
The equilibrium concentrations of the atomic form of each element at
Concept Introduction:
Equilibrium constant
In gas phase reactions, partial pressure is used to write equilibrium equation than molar concentration. Equilibrium constant
Consider the reaction where A reacts to give B.
On rearranging,
Where,
(a)
Explanation of Solution
Using ICE table to calculate x value.
The concentration of
At equilibrium,
X value is calculated as,
Taken an example of the Bromine,
From the above same method is used to other elements to get equilibrium concentrations of the atomic form of each element at
Hence, the equilibrium concentrations of the atomic form of each element at
(b)
Interpretation:
The element which has lowest
(b)
Explanation of Solution
In generally, a molecule which one have lowest enthalpy value, it is give a more product so the enthalpy vales of given molecules are,
The calculated equilibrium concentrations of the atomic form of each element at
From the above, data the lowest enthalpy containing molecule is yields more product.
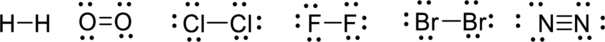
From the above, Lewis structures and concentrations of elements, the single bonded compound forms more products.
Want to see more full solutions like this?
Chapter 12 Solutions
Chemistry: The Molecular Science
- Write an equation for an equilibrium system that would lead to the following expressions (ac) for K. (a) K=(Pco)2 (PH2)5(PC2H6)(PH2O)2 (b) K=(PNH3)4 (PO2)5(PNO)4 (PH2O)6 (c) K=[ ClO3 ]2 [ Mn2+ ]2(Pcl2)[ MNO4 ]2 [ H+ ]4 ; liquid water is a productarrow_forwardAt a certain temperature, K=0.29 for the decomposition of two moles of iodine trichloride, ICl3(s), to chlorine and iodine gases. The partial pressure of chlorine gas at equilibrium is three times that of iodine gas. What are the partial pressures of iodine and chlorine at equilibrium?arrow_forwardShow that the complete chemical equation, the total ionic equation, and the net ionic equation for the reaction represented by the equation KI(aq)+I2(aq)KI3(aq) give the same expression for the reaction quotient. KI3 is composed of the ions K+ and I3-.arrow_forward
- At 454 K, Al,Cl6(g) reacts to form Al;Cl,(g) according to the equation 3 Al,Cl,(g) = 2 Al;Cl,(g) In an experiment at this temperature, the equilibrium par- tial pressure of Al,Cl(g) is 1.00 atm and the equilibrium partial pressure of Al;Cl,(g) is 1.02 × 10-2 atm. Compute the equilibrium constant of the preceding reaction at 454 K.arrow_forwardBe sure to answer all parts.The equilibrium constant (K) for the formation of nitrosyl chloride, an orange-yellow compound, from nitric oxide and molecular chlorine 2NO(g) + Cl2(g) = 2NOCI(g) is 4 x 10° at a certain temperature. In an experiment, 2.60 × 102 mole of NO, 2.40 × 103 mole of Cl2, and 4.60 moles of NOCI are mixed in a 2.80–L flask. What is Q. for the experiment? х 10 (Enter your answer in scientific notation.) In which direction will the system proceed to reach equilibrium? The reaction will proceed to the left. The reaction will proceed to the right. The reaction is at equilibrium.arrow_forwardPhosphorus pentachloride is formed when phosphorus trichloride and chlorine react according to the following reaction. PCI3(g)+ Cl2(g) = PCl5(g) The equilibrium constant for the reaction is Kc, = 37.5 at 145 °C. If 0.222 mol of phosphorus trichloride is added to 0.750 mol of chlorine in a 1.69 - L reaction vessel and allowed to react at this temperature, what is the equilibrium concentration (in mol/L) of phosphorus pentachloride? Report your answer to THREE significant figures.arrow_forward
- Suppose a 250. mL flask is filled with 1.2 mol of Br₂, 0.50 mol of BrOCl and 1.9 mol of BrC1. The following reaction becomes possible: Br₂(g) + OC1₂(g) → BrOC1 (g) + BrC1 (g) The equilibrium constant K for this reaction is 0.156 at the temperature of the flask. Calculate the equilibrium molarity of OC12. Round your answer to two decimal places. |] M X Ś ?arrow_forwardThe equilibrium constant, Ke, for the following reaction is 9.52x102 at 350 K. CH4 (g) + CC14 (g)=2 CH2Cl, (g) Calculate the equilibrium concentrations of reactants and product when 0.263 moles of CH4 and 0.263 moles of CCl, are introduced into a 1.00 L vessel at 350 K. [ CH4] [ CC4 ] [ CH,Cl2 ] = | M M Marrow_forwardWrite the equilibrium constant expression, K, for the following reaction. Please enter the compounds in the order given in the reaction. If either the numerator or denominator is blank, please enter 1 HNO2 (aq) + H2O(1)=H30*(aq) + NO2¯ (aq) K =arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





