
Concept explainers
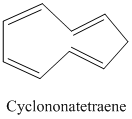
Evaluate each of the following processes applied to cyclononatetraene, and decide whether
the species formed is
Addition of one more π electron, to give
Addition of two more π electrons, to give
Loss of H+ from the
Loss of H+ from one of the
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry - Standalone book
- Aromatic compounds must have a p orbital on every atom in the ring. Define this ?arrow_forwardSpecify whether the two structures are resonance contributors to the same resonance hybrid. Be sure to explain your reasoning. If yes, be sure to specify which is preferred and why. H2C=NH2arrow_forwardCompare the structures of 1,4-pentadiene and divinylamine: 1,4-Pentadiene The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using this information, identify the hybridization state of the nitrogen atom in divinylamine. O sp³ hybridized O sp³d hybridized sp² hybridized O sp hybridized N H Divinylamine Justify your answer. The lone pair is a part of the TT system in the second compound. Therefore the compound is conjugated ✓ .As such, the compound absorbs light above 200 nm (UV light). In contrast, 1,4-pentadiene has two double bonds and therefore does not absorb UV light in the region between 200 and 400 nm. 3 isolated cumulated conjugatedarrow_forward
- Which of the following structures is/are not aromatic? 3 O 2 and 3 only O 1 and 3 only O 1 and 2 only O 2 only O 3 only 2. 1-arrow_forwardn of the following molecules could be considered aromatic? Assume all of the compounds are planar. T: Are these proper Lewis structures? ZI NH₂ N HBarrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energyarrow_forward
- The pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energy. (continued)762 CHAPTER 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy(e) Show how many electrons are in each MO for the pentadienyl radical (ground state).(f) Show how your molecular orbital picture agrees with the resonance picture showing delocalization of the unpairedelectron onto three carbon atoms.(g) Remove the highest-energy electron from the pentadienyl radical to give the pentadienyl cation. Which carbon atomsshare the positive charge? Does this picture agree with the resonance picture?(h) Add an…arrow_forwardSketch the ultraviolet absorption spectrum of 1,3-Butadiene, matching peaks to electronic transitions. show that four isolated p orbital on carbon atoms combine to form pi orbitalarrow_forwardChoose the BEST classification for each structure shown below. Classifications to Choose from: Alkane, Alkene, Alkyne, Aromatic, Organic Halide (Alkyl Halide), Ketone, Aldehyde, Carboxylic Acid, Ether, Ester, Amine, Amide, Alcohol, Nitrile. CH3 CH3-CH2-CH2-CH2-CH2-CH3 нн H CH2 CH-C Н-с-с-о-с-н OH н H CH2 Structure 1 Structure 2 Structure 3 Structure 4 NH2 CH3 `CH CH3 CH2 CH3 „CH CH3 CH3 `CH2 `CH2 CH3 Structure 7 Structure 5 Structure 6 Structure 8 H CH3 -N- -CH3 F- „CH2-CH2-CH3 CH2 CH3 F Structure 9 Structure 10 Structure 11 Structure 12 o=Uarrow_forward
- Which structure is NOT aromatic? CH CHarrow_forwardHow many π molecular orbitals (π-MOs) are formed from the linear combination of atomicorbitals (AOs) contributing to the π-bonding in pentene?arrow_forwardSelect all statements that are true regarding tertiary radicals - Tertiary radicals are more stable than primary radicals due to increased hyperconjugation from attached alkyl groups. - The CH bond that leads to a tertiary radical via homolysis has a higher bond dissociation energy that the CH bond leading to a primary radical - Tertiary radical hybridization geometry is best described as sp2 hybridization rather than sp3 hybridization - Radical chlorination reactions are more regioselective than radical bromination reactions when a tertiary radical can form.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
