
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 35P
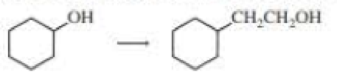
Using the given starting material, any necessary inorganic reagent, and any carbon-containing compounds with no more than two carbons, indicate how each of the following compounds con be prepared:

Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which of the following is an acetal?
H3C
H;C
но
CH3
CH3
H3C-
H3C
H3C
H3C
OH
А
C
A and C
B and D
A
O A, B, and D
D
B.
Identify the correct reagent for each of the following reactions.
Identify the best reagents to complete the following
reaction.
CI
o
Chapter 12 Solutions
Organic Chemistry
Ch. 12.1 - Prob. 1PCh. 12.2 - Which is more reactive an organolithium compound...Ch. 12.2 - Prob. 3PCh. 12.3 - PROBLEM 6♦
Explain why tertiary alkyl halides...Ch. 12.3 - Muscalure is the sex attractant of the common...Ch. 12.3 - Prob. 8PCh. 12.3 - Prob. 9PCh. 12.3 - Prob. 10PCh. 12.4 - Prob. 13PCh. 12.4 - Prob. 14P
Ch. 12.4 - Prob. 15PCh. 12.4 - Show how the Suzuki and/or Heck reactions can be...Ch. 12.4 - Identify two pairs of an alkyl bromide and an...Ch. 12.5 - Prob. 19PCh. 12.5 - Draw the product of ring-closing metathesis for...Ch. 12.5 - Prob. 22PCh. 12 - Prob. 23PCh. 12 - Prob. 24PCh. 12 - Identify A through H.Ch. 12 - 26. Using the given starting material, any...Ch. 12 - Prob. 27PCh. 12 - Prob. 28PCh. 12 - Prob. 29PCh. 12 - Using ethynyleyclohexane as a starting material...Ch. 12 - Prob. 31PCh. 12 - Using the given starting material, any necessary...Ch. 12 - Prob. 33PCh. 12 - A student added an equivalent of...Ch. 12 - Using the given starting material, any necessary...Ch. 12 - Prob. 36PCh. 12 - Prob. 37PCh. 12 - Bombykol is the sex pheromone of the silk moth....Ch. 12 - Prob. 39PCh. 12 - A dibromide loses only one bromine when it reacts...Ch. 12 - What starting material is required in order to...Ch. 12 - Prob. 42PCh. 12 - Prob. 1PCh. 12 - Prob. 2PCh. 12 - Prob. 3PCh. 12 - Prob. 4P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous OH?arrow_forwardUsing the given starting material, any necessary inorganic reagents, and any carbon-containing compounds with no more than two carbons, indicate how each of the following compounds can be prepared:arrow_forwardDraw the six products (including stereoisomers) formed when A is treated wit NBS + hv.arrow_forward
- What is the IUPAC name of the following compound? CH;CH=CCH2CH2OH (A) 3-Phenylpent-3-enol B 3-Phenylpent-2-enol C 3-Benzenepent-3-enol D 3-Phenylpent-2-en-5-ol E 5-Hydroxy-3-phenylpent-2-enearrow_forwardprovide the missing reagents as appropriatearrow_forwardMelamine, used as a fire retardant and a component of the writing surface of white boards, can be prepared from s-trichlorotriazine through a series of SNAr reactions with ammonia. The first substitution takes place rapidly at room temperature. The second substitution takes place near 100 °C, and the third substitution requires even higher temperature and pressure. Provide an explanation fatr this reactivity.arrow_forward
- Draw the correct organic product of the following oxidation reaction:arrow_forwardSynthesize each compound from cyclohexanol, ethanol, and any other needed reagents.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardAcyclovir is an effective antiviral agent used to treat the herpes simplexvirus. (a) Draw the enol form of acyclovir, and explain why it is aromatic.(b) Why is acyclovir typically drawn in its keto form, despite the fact thatits enol is aromatic?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY