
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.66AQAP
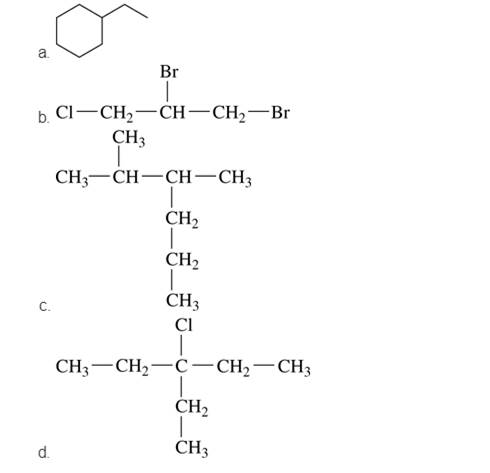
Give the IUPAC name for each of the following: (12.3)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The representation of a one-dimensional velocity distribution function for a gas, as the temperature increases:a) it becomes more flattenedb) the maximum occurs for vi = 0 m/sExplain it.
The velocity distribution function of gas moleculesa) is used to measure their velocity, since the small size of gas molecules means that it cannot be measured in any other wayb) is only used to describe the velocity of particles if their density is very high.c) describes the probability that a gas particle has a velocity in a given interval of velocities
Explain why in the representation of a one-dimensional velocity distribution function for a particular gas, the maximum occurs for vi = 0 m/s.
Chapter 12 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 12.1 - Identify each of the following as a formula of an...Ch. 12.1 - Prob. 12.2QAPCh. 12.1 - Prob. 12.3QAPCh. 12.1 - Prob. 12.4QAPCh. 12.1 - Prob. 12.5QAPCh. 12.1 - Prob. 12.6QAPCh. 12.1 - Prob. 12.7QAPCh. 12.1 - Prob. 12.8QAPCh. 12.2 - Give the IUPAC name for each of the following...Ch. 12.2 - Prob. 12.10QAP
Ch. 12.2 - Prob. 12.11QAPCh. 12.2 - Prob. 12.12QAPCh. 12.3 - Prob. 12.13QAPCh. 12.3 - Prob. 12.14QAPCh. 12.3 - Give the IUPAC name for each of the following:Ch. 12.3 - Give the IUPAC name for each of the following:Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the line-angle structural formula for each of...Ch. 12.3 - Draw the line-angle structural formula for each of...Ch. 12.4 - Heptane, used as a solvent for rubber cement, has...Ch. 12.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 12.4 - Prob. 12.23QAPCh. 12.4 - Prob. 12.24QAPCh. 12.4 - Prob. 12.25QAPCh. 12.4 - Prob. 12.26QAPCh. 12.5 - Prob. 12.27QAPCh. 12.5 - Prob. 12.28QAPCh. 12.5 - Prob. 12.29QAPCh. 12.5 - Prob. 12.30QAPCh. 12.5 - Prob. 12.31QAPCh. 12.5 - Prob. 12.32QAPCh. 12.6 - 12.33 Give the IUPAC name for each of the...Ch. 12.6 - Prob. 12.34QAPCh. 12.6 - Prob. 12.35QAPCh. 12.6 - Prob. 12.36QAPCh. 12.6 - Prob. 12.37QAPCh. 12.6 - Prob. 12.38QAPCh. 12.7 - Prob. 12.39QAPCh. 12.7 - Prob. 12.40QAPCh. 12.7 - Prob. 12.41QAPCh. 12.7 - Prob. 12.42QAPCh. 12.7 - Prob. 12.43QAPCh. 12.7 - Prob. 12.44QAPCh. 12.7 - Prob. 12.45QAPCh. 12.7 - Prob. 12.46QAPCh. 12.8 - Prob. 12.47QAPCh. 12.8 - Give the IUPAC name and any common name for each...Ch. 12.8 - Prob. 12.49QAPCh. 12.8 - Prob. 12.50QAPCh. 12.8 - Prob. 12.51QAPCh. 12.8 - Prob. 12.52QAPCh. 12.8 - Prob. 12.53QAPCh. 12.8 - Prob. 12.54QAPCh. 12 - Prob. 12.55UTCCh. 12 - Prob. 12.56UTCCh. 12 - Prob. 12.57UTCCh. 12 - Prob. 12.58UTCCh. 12 - Prob. 12.59UTCCh. 12 - Prob. 12.60UTCCh. 12 - Prob. 12.61UTCCh. 12 - Prob. 12.62UTCCh. 12 - Give the number of carbon atoms and the types of...Ch. 12 - Prob. 12.64UTCCh. 12 - Prob. 12.65AQAPCh. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Prob. 12.67AQAPCh. 12 - Prob. 12.68AQAPCh. 12 - Prob. 12.69AQAPCh. 12 - 12.70 Give the IUPAC name for each of the...Ch. 12 - Prob. 12.71AQAPCh. 12 - Prob. 12.72AQAPCh. 12 - Prob. 12.73AQAPCh. 12 - Prob. 12.74AQAPCh. 12 - Prob. 12.75AQAPCh. 12 - Prob. 12.76AQAPCh. 12 - Prob. 12.77AQAPCh. 12 - Prob. 12.78AQAPCh. 12 - Prob. 12.79AQAPCh. 12 - Prob. 12.80AQAPCh. 12 - Prob. 12.81AQAPCh. 12 - Prob. 12.82AQAPCh. 12 - Prob. 12.83AQAPCh. 12 - Prob. 12.84AQAPCh. 12 - Prob. 12.85AQAPCh. 12 - Prob. 12.86AQAPCh. 12 - Prob. 12.87AQAPCh. 12 - Prob. 12.88AQAPCh. 12 - Prob. 12.89CQCh. 12 - Prob. 12.90CQCh. 12 - Prob. 12.91CQCh. 12 - Prob. 12.92CQCh. 12 - Prob. 12.93CQCh. 12 - Prob. 12.94CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain why the representation of a one-dimensional velocity distribution function for a particular gas becomes flatter as the temperature increases.arrow_forwardDraw a Lewis structure for each of the following molecules and assign charges where appropriate. The order in which the atoms are connected is given in parentheses. a. CIFCIF b. BrCNBrCN 0 c. SOCI2 × (CISCIO) SOC₁₂ (CISCI) You can draw both an octet and a valence shell expanded structure. Considering the following structural information, which is the better one: The measured S-OS-O bond length in SOC12SOCl2 is 1.43 Å. For comparison, that in SO2SO2 is 1.43 Å [Exercise 1-9, part (b)], that in CHзSOHCH3 SOH d. CH3NH2CH3NH2 (methanesulfenic acid) is 1.66 A. e. CH3OCH3 CH3 OCH3 NH2 f. N2H2× (HNNH) N2 H2 (HNNH) g. CH2COCH₂ CO h. HN3× (HNNN) HN3 (HNNN) i. N20 × (NNO) N2O (NNO)arrow_forwardbre The reaction sequence shown in Scheme 5 demonstrates the synthesis of a substituted benzene derivative Q. wolsd works 2 NH2 NaNO2, HCI (apexe) 13× (1 HNO3, H2SO4 C6H5CIN2 0°C HOTE CHINO₂ N O *O₂H ( PO Q Я Scheme 5 2 bag abouoqmics to sounde odi WEIC (i) Draw the structure of intermediate O. [2 marks] to noitsmot od: tot meinedogm, noit so oft listsb ni zaupaib bas wa (ii) Draw the mechanism for the transformation of aniline N to intermediate O. Spoilage (b) [6 marks] (iii) Identify the reagent X used to convert compound O to the iodinated compound [tom E P. vueimado oilovonsa ni moitos nolisbnolov ayd toes ai tedw nisiqx (iv) Identify the possible structures of compound Q. [2 marks] [2 marks] [shom 2] (v) bus noires goiribbeolovo xnivollot adj to subora sidab Draw the mechanism for the transformation of intermediate P to compound Q. [5 marks] vi (vi) Account for the regiochemical outcome observed in the reaction forming compound Q. [3 marks]arrow_forward
- Please correct answer and don't used hand raitingarrow_forwardThe vibrational contribution isa) temperature independent for internal energy and heat capacityb) temperature dependent for internal energy and heat capacityc) temperature independent for heat capacityd) temperature independent for internal energyarrow_forwardQuantum mechanics. Explain the basis of approximating the summation to an integral in translational motion.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY