
(a)
Interpretation:
The Lewis structure for given acetone and carbon disulfide molecules should be determined and the deviation from ideal behavior should be explained using intermolecular forces.
Concept Introduction:
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
- Sometimes the
chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance.
Lewis structure for any molecule is drawn by using the following steps,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
Intermolecular force: The attractive force that withholds two molecules is called as intermolecular force. The influence of intermolecular forces depends on molar mass and the functional group present in the molecule.
Types and strength of intermolecular forces in decreasing order:
Polar molecule: Polar molecules have large dipole moments.
Non-Polar molecules: Non-polar molecules have bonded atoms with similar electronegativity results to have zero dipole moments.
(a)
Explanation of Solution
The Lewis structures are as follows,
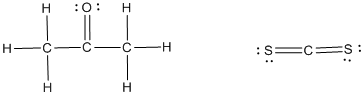
The given molecule acetone is polar whereas the molecule carbon disulfide is nonpolar molecule.
According to Like dissolves like principle, the attraction between acetone and
Due to weak attractions the molecules will leave the solution that result in increased vapor pressure of the solution than the sum of vapor pressures by Raoutl’s law.
(b)
Interpretation:
The vapor pressure of the solution under ideal condition should be determined.
Concept Introduction:
Raoult’s law: Raoult’s law states that solvent partial pressure over a solution is equal to vapor pressure of pure solvent multiplied with mole fraction of the solvent.
Where,
The solution that obeys Raoult’s law is defined as ideal solution.
(b)
Explanation of Solution
The ideal solution should obey Raoult’s law.
Partial pressure for A is as follows,
Partial pressure for B is as follows,
The ideal pressure of the solution is
(c)
Interpretation:
The sign of
Concept Introduction:
Enthalpy
Enthalpy is the amount energy absorbed or released in a process.
The enthalpy change in a system
Where,
(c)
Explanation of Solution
The given molecule acetone is polar whereas the molecule carbon disulfide is nonpolar molecule.
According to Like dissolves like principle, the attraction between acetone and
Due to weak attractions the molecules will leave the solution that result in increased vapor pressure of the solution than the sum of vapor pressures by Raoutl’s law.
Therefore, the solution deviates positively from Raoutl’s law which says that heat of solution is positive that means mixing is an endothermic process (heat is absorbed by the system).
Want to see more full solutions like this?
Chapter 12 Solutions
Chemistry
- To make homemade ice cream, you cool the milk and cream by immersing the container in ice and a concentrated solution of rock salt (NaCl) in water. If you want to have a water-salt solution that freezes at 10. C, what mass of NaCl must you add to 3.0 kg of water? (Assume the vant Hoff factor, i, for NaCl is 1.85.)arrow_forwardA 0.029 M solution of potassium sulfate has an osmotic pressure of 1.79 atm at 25 C. (a) Calculate the vant Hoff factor, i, for this solution. (b) Would the vant Hoff factor be larger, smaller, or the same for a 0.050 M solution of this compound?arrow_forwardWhat is the usual solubility behavior of an ionic compound in water when the temperature is raised? Give an example of an exception to this behavior.arrow_forward
- A compound contains 42.9% C, 2.4% H, 16.6% N, and 38.1% O. The addition of 3.16 g of this compound to 75.0 mL of cyclohexane (d=0.779g/mL) gives a solution with a freezing point at 0.0C. Using Table 10.2, determine the molecular formula of the compound.arrow_forwardCalculate the freezing point and normal boiling points of each of the following aqueous solutions. (a) 2.63 m acetic acid (b) 33.0 % by mass lactose, C12H22O11 (c) 32.15 mL of ethylene glycol, C2H6O2(d=1.113g/mL) in 624 mL of water (d=1.00g/mL)arrow_forwardIce Cream A rock salt (NaCl), ice, and water mixture isused to cool milk and cream to make homemade icecream. How many grams of rock salt must be added towater to lower the freezing point by 10.0°C?arrow_forward
- The freezing point of a 0.031-m solution of copper(II) sulfate in water is 0.075 C. (a) Calculate the vant Hoff factor, i, for this solution. (b) Would the vant Hoff factor be larger, smaller, or the same for a 0.050-m solution of this compound?arrow_forwardSilver ion has an average concentration of 28 ppb (parts per billion) in U.S. water supplies. (a) What is the molality of the silver ion? (b) If you wanted 1.0 102 g of silver and could recover it chemically from water supplies, what volume of water in liters would you have to treat? (Assume the density of water is 1.0g/cm3.)arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





