
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.9, Problem DQ
Interpretation Introduction
Interpretation:
The relative amount of trans-tetrol to cis-tetrol at
Concept Introduction:
Ring opening: Ring opening of epoxide takes place by acid-catalyzed hydrolysis of an epoxide.
Acid-catalyzed hydrolysis of an epoxide via
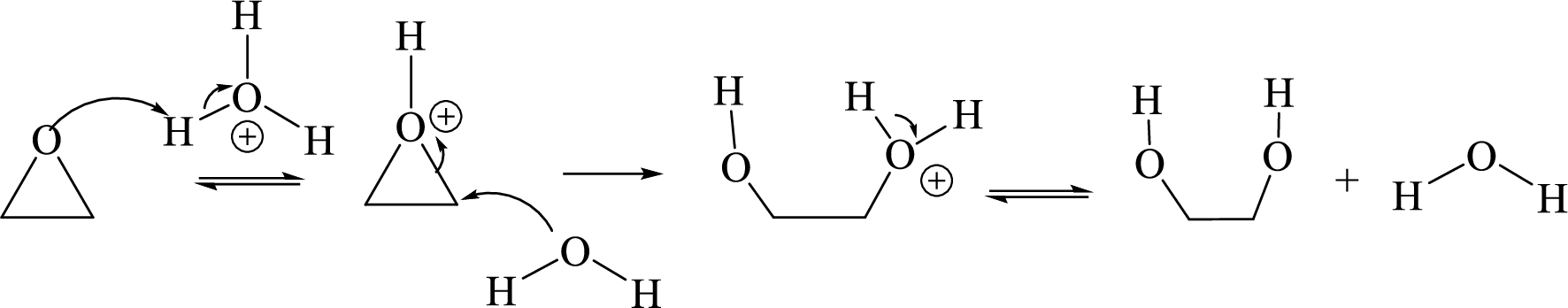
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10. Explain the reaction shown below. Include the following pieces in your explanation.
Identify the electrophile and the nucleophile.
Draw a curved arrow mechanism for this reaction. (Assume an acidic work-up).
Explain the electron flow.
Indicate any regiochemical or stereochemical preferences.
one
b.
c.
d.
LIAIH4
OH
one
Draw the structure(s) of the major organic product(s) of the following reaction.
H,C.
1. NaOCH3 / CH3OH
2. 1 eq. 2-bromo-2-methylpropane
• You do not have to consider stereochemistry.
• Omit products derived from the acidic or basic reagent itself, e.g. HN(i-Pr)2 derived from N(i-Pr)2.
• If no reaction occurs, draw the organic starting material.
• If substantial starting material is present at the end of the reaction, include it in the products.
• Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
Separate structures with + signs from the drop-down menu.
16. Explain the reaction shown below. Include the following pieces in your explanation.
Identify the electrophile and the nucleophile.
Draw a curved arrow mechanism for this reaction. (Assume an acidic work-up).
Explain the electron flow.
Indicate any regiochemical or stereochemical preferences.
Ph-0
a.
b.
c.
d.
Ph
OH
Chapter 11 Solutions
Organic Chemistry
Ch. 11.2 - Write IUPAC and common names for these ethers. (a)...Ch. 11.3 - Arrange these compounds in order of increasing...Ch. 11.4 - Show how you might use the Williamson ether...Ch. 11.4 - Show how ethyl hexyl ether might be prepared by a...Ch. 11.5 - Account for the fact that treatment of tert-butyl...Ch. 11.5 - Draw structural formulas for the major products of...Ch. 11.6 - Prob. 11.7PCh. 11.8 - Draw the expected products of Sharpless...Ch. 11.9 - Prob. AQCh. 11.9 - Prob. BQ
Ch. 11.9 - Prob. CQCh. 11.9 - Prob. DQCh. 11 - Write names for these compounds. Where possible,...Ch. 11 - Prob. 11.11PCh. 11 - Each compound given in this problem is a common...Ch. 11 - Account for the fact that tetrahydrofuran (THF) is...Ch. 11 - Prob. 11.14PCh. 11 - Write equations to show a combination of reactants...Ch. 11 - Propose a mechanism for this reaction.Ch. 11 - Prob. 11.17PCh. 11 - Prob. 11.18PCh. 11 - Prob. 11.19PCh. 11 - Prob. 11.20PCh. 11 - Ethylene oxide is the starting material for the...Ch. 11 - Prob. 11.22PCh. 11 - Predict the structural formula of the major...Ch. 11 - The following equation shows the reaction of...Ch. 11 - Propose a mechanism to account for this...Ch. 11 - Acid-catalyzed hydrolysis of the following epoxide...Ch. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Prob. 11.29PCh. 11 - Propose a mechanism for the following...Ch. 11 - Show reagents and experimental conditions to...Ch. 11 - Starting with cis-3-hexene, show how to prepare...Ch. 11 - Show reagents to convert cycloheptene to each of...Ch. 11 - Show reagents to convert bromocyclopentane to each...Ch. 11 - Prob. 11.35PCh. 11 - Starting with acetylene and ethylene oxide as the...Ch. 11 - Following are the steps in the industrial...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Aldehydes and ketones react with one molecule of...Ch. 11 - Prob. 11.42PCh. 11 - Write the products of the following sequences of...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - During the synthesis of the antiasthmatic drug...Ch. 11 - Prob. 11.48P
Knowledge Booster
Similar questions
- Choose all correct statements. 1. Gilman reagents react with alkyl halides, including vinyl and aryl halides. 2. Grignard reagents react with alkyl halides, including vinyl and aryl halides. 3. Deprotonation of an alcohol group forms an oxonium intermediate. 4. Whenever a neutral nucleophile attacks, a proton transfer step follows. 5. An alcohol that is chiral at the a-carbon will undergo inversion of absolute configuration when it is converted into an alkyl tosylate. C 1 2 3 4arrow_forwardC. Draw the structure for the product formed in each of step of the following synthetic sequences. 1. C NH₂ SOCI₂ or P₂O5 or POCI3 (dehydration) 1.CH3MgBr 2.H3O+ 1. NaOH, 12 2. H3O+ + yellow precipitatearrow_forwardWhich condition or sequence is ideal for converting ethyne into the epoxide shown below? A. 1. D2 (1 equivalent)/Pd-C 2. H3CC(O)OOH B. 1. D2/Lindlar 2. H3CC(O)OOH C. 1. H3CC(O)OOH 2. D2/ Pd-C D. 1. Na/ND3 2. H3CC(O)OOHarrow_forward
- Acyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. H3C. ... NH2 HCI/H₂O reflux • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • Do not include counter-ions, e.g., Na+, I, in your answer. • In cases where there is more than one answer, just draw one. + 4 n [ ?arrow_forwardHow does increasing base, solvent, or alkyl halide or aptly halide + base affect or decreasing them affect the reaction rate for e2/e1 and sn2/sn1? Please provide examplesarrow_forward10. Explain the reaction shown below. Include the following pieces in your explanation. a. Identify the electrophile and the nucleophile. b. Draw a curved arrow mechanism for this reaction. (Assume an acidic work-up). Explain the electron flow. Indicate any regiochemical or stereochemical preferences. C. d. LIAIH4 OHarrow_forward
- I. Propose a one-step synthetic route to get the following compound using only SN2. Explain. H3C- CH3 H3C II. Propose a reasonable mechanism to explain the following reaction. Explain. Br etanol calorarrow_forward8 Organocuprates predominantly react to give 1,4-addition products with a,B-unsatu- rated carbonyl species, while Grignard reagents often add to the carbonyl, in a process referred to as 1,2-addition. To increase the yield of 1,4-addition products, Cul is added to convert an easily prepared Grignard reagent into a organocuprate reagent in situ (during the reaction). Predict the major product and stereochemistry of the following reaction, assuming that the more stable chair product predominates. MgBr Cul, THF, -30 C 2. Equilibration to more stable chair product in basearrow_forwardChoose the reagent(s) that would be most likely to complete this reaction. I||| / A B C D 1. BH3-THF 2. H2O2, NaOH Br₂ H₂O OsO4 (catalytic) NMO RCO3H Donearrow_forward
- 20. a. Label the reactive features, highlight the most reactive one, then highlight what it needs. Also, state if the reaction will start to create a carbocation, carbon radical, or carbanion, or will cause loss of aromatic character. If a carbocation, carbon radical, or carbanion starts to develop, label where that will occur. o-cresol + 2-methylpropanoyl chloride + AICI3 b. Use mechanism arrows to illustrate the reaction that occurs. c. If applicable, use stabilization resources to deal with the carbocation, carbon radical, or carbanion that starts to develop during the reaction, and draw the structure of any resonance-stabilized intermediate. d. Continue labeling and diagramming the reaction until you find the major stable product(s). e. Finally, state the stereochemistry of the major product(s) and use either Fisher projection or perspective formula representations to illustrate that stereochemistry.arrow_forward15.Which of the following correctly explains why alkyl groups are ortho/para-directing groups? a. Meta substitution results in an arenium ion in which a positive charge is destabilized by an adjacent electron-withdrawing group. b. Ortho/para substitution results in an arenium ion intermediale in which all atoms have complete octets. c. A lone pair of electrons on the alkyl group participates in re sonance and stabilizes the arenium ion intermediate in ortho/para substitution. d. The most stable resonance structure for the arenium ion intermediate in ortho/para substitution contains a 2° carbocation. e. The alkyl group is electron donating via induction and stabilizes the adjacent positive charge in the ortho/para substitution intermediate. 5.arrow_forwardk. I. m. n. 0. р. q. r. Hзвод Ph H3C H3C H3C H3C H3C H3C Phi OTS CH3 ОН CH3 OSO4 NMO CH3 CH3 CH3 ОН -CH3 t-BuOK t-BuOH CH3 1) NaNH2 2) Ph HBr peroxides H2 Lindlar catalyst Br 1) BH3, THF 2) H2O2, aq. NaOH 1) NaH 2) CHzl HgSO4 Н300 H₂SO4 Дarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT