
An excellent way to make highly pure nickel metal for use in specialized steel alloys is to decompose Ni(CO)4 by heating it in a vacuum to slightly above room temperature.
Ni(CO)4(g) → Ni(s) + 4 CO(g)
The reaction is proposed to occur in four steps, the first of which is
Ni(CO)4(g) → Ni(CO)3(g) + CO(g)
Kinetic studies of this first-order decomposition reaction have been carried out between 47.3 °C and 66.0 °C to give the results in the table.*
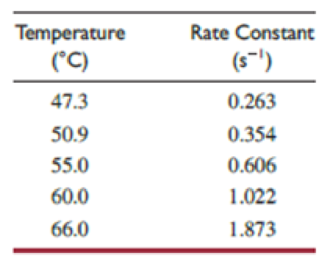
- (a) Determine the activation energy for this reaction.
- (b) Ni(CO)4 is formed by the reaction of nickel metal with carbon monoxide. Suppose that 2.05 g CO is combined with 0.125 g nickel metal. Determine the maximum mass (g) of Ni(CO)4 that can be formed.
Replacement of CO by another molecule in Ni(CO)4 was studied in the nonaqueous solvents toluene and hexane to understand the general principles that govern the chemistry of such compounds.*
Ni(CO)4(g) + P(CH3)3 → Ni(CO)3P(CH3)3 + CO
A detailed study of the kinetics of the reaction led to the mechanism
- (c) Which step in the mechanism is unimolecular? Which is bimolecular?
- (d) Add the steps of the mechanism to show that the result is the balanced equation for the observed reaction.
- (e) Is there an intermediate in this reaction? If so, what is it?
- (f) It was found that doubling the concentration of Ni(CO)4 increased the reaction rate by a factor of 2. Doubling the concentration of P(CH3)3 had no effect on the reaction rate. Based on this information, write the rate equation for the reaction.
- (g) Does the experimental rate equation support the proposed mechanism? Why or why not?
(a)
Interpretation:
The activation energy of the reaction has to be determined. The reaction is given below.
Concept Introduction:
The Arrhenius equation is given below.
Where,
By taking
The above equation is in the form of
Explanation of Solution
A table has to be made as shown below by using the given data.
A graph can be plotted as
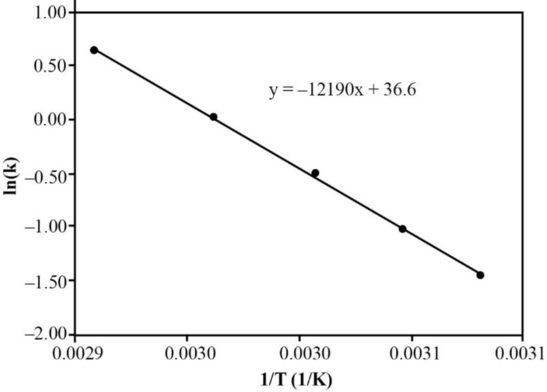
Figure
From the slope of the above graph, the activation energy of the reaction can be calculated.
Therefore, the activation energy of the reaction is
(b)
Interpretation:
The maximum mass
Explanation of Solution
The reverse reaction of the given reaction represents the formation of
Four moles of
Determination of limiting reagent:
The number of
The mass of
Therefore, the maximum mass
(c)
Interpretation:
The unimolecular and bimolecular step have to be chosen from the given mechanism.
Explanation of Solution
A reaction is called as elementary and unimolecular reaction if the reaction involves only one reactant. If the reaction has exactly two reactants, either two of the same reactants or one of each of different reactants, then the reaction is bimolecular and elementary.
The first step is unimolecular as it involves exactly one reactant. The second step is bimolecular as it involves exactly two reactants.
(d)
Interpretation:
The steps of the given mechanism has to be added in order to show that the result is the balanced equation for the observed reaction.
Explanation of Solution
The given mechanisms can be added as given below.
The resulting equation is the same as that given for the observed reaction.
(e)
Interpretation:
The intermediate involved in the reaction has to be determined.
Explanation of Solution
An intermediate is a chemical substance that is generated in an early step of a reaction and then used up in a later step. In the given mechanism, the intermediate is
(f)
Interpretation:
Doubling the concentration of
Explanation of Solution
The reaction is given below.
Suppose the rate of the above reaction is as follows,
Where,
Given that, doubling the concentration of
The second condition is doubling the concentration of
Now, the rate equation can be written as shown below.
(g)
Interpretation:
The experimental rate equation support the proposed mechanism or not, has to be explained. The mechanism is given below.
Explanation of Solution
The experimental rate equation can be written as shown below.
The slowest step in a mechanism is the rate determining step. Thus, according to the mechanism, the rate determining step is the first step. The rate equation can be written as follows,
Thus, the experimental rate equation supports the proposed mechanism.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry: The Molecular Science
- Nitrosyl bromide, NOBr, is formed from NO and Br2: 2 NO(g) + Br2(g) 2 NOBr(g) Experiments show that this reaction is second-order in NO and first-order in Br2. (a) Write the rate equation for the reaction. (b) How does the initial reaction rate change if the concentration of Br2 is changed from 0.0022 mol/L to 0.0066 mol/L? (c) What is the change in the initial rate if the concentration of NO is changed from 0.0024 mol/L to 0.0012 mol/L?arrow_forwardIodomethane (CH3I) is a commonly used reagent in organic chemistry. When used properly, this reagent allows chemists to introduce methyl groups in many different useful applications. The chemical does pose a risk as a carcinogen, possibly owing to iodomethanes ability to react with portions of the DNA strand (if they were to come in contact). Consider the following hypothetical initial rates data: [DNA]0 ( mol/L) [CH3I]0 ( mol/L) Initial Rate (mol/Ls) 0.100 0.100 3.20 104 0.100 0.200 6.40 104 0.200 0.200 1.28 103 Which of the following could be a possible mechanism to explain the initial rate data? MechanismIDNA+CH3IDNACH3++IMechanismIICH3ICH3++ISlowDNA+CH3+DNACH3+Fastarrow_forwardIsomerization of CH3NC occurs slowly when CH3NC is heated. CH3NC(g) CH3CN(g) To study the rate of this reaction at 488 K, data on [CH3NC] were collected at various times. Analysis led to the following graph. (a) What is the rate law for this reaction? (b) What is the equation for the straight line in this graph? (c) Calculate the rate constant for this reaction. (d) How long does it take for half of the sample to isomerize? (e) What is the concentration of CH3NC after 1.0 104 s?arrow_forward
- Ozone, O3, in the Earths upper atmosphere decomposes according to the equation 2 O3(g) 3 O2(g) The mechanism of the reaction is thought to proceed through an initial fast, reversible step followed by a slow, second step. Step 1: Fast, reversible O3(g) O2(g) + O(g) Step 2: Slow O3(g) + O(g) 2 O2(g) (a) Which of the steps is rate-determining? (b) Write the rate equation for the rate-determining steparrow_forwardAt 500 K in the presence of a copper surface, ethanol decomposes according to the equation C2H5OH(g)CH3CHO(g)+H2(g) The pressure of C2H5OH was measured as a function of time and the following data were obtained: Time(s) PC2H5OH(torr) 0 250. 100. 237 200. 224 300. 211 400. 198 500. 185 Since the pressure of a gas is directly proportional to the concentration of gas, we can express the rate law for a gaseous reaction in terms of partial pressures. Using the above data, deduce the rate law, the integrated rate law, and the value of the rate constant, all in terms of pressure units in atm and time in seconds. Predict the pressure of C2H5OH after 900. s from the start of the reaction. (Hint: To determine the order of the reaction with respect to C2H5OH, compare how the pressure of C2H5OH decreases with each time listing.)arrow_forwardAt 573 K, gaseous NO2(g) decomposes, forming NO(g) and O2(g). If a vessel containing NO2(g) has an initial concentration of 1.9 102 mol/L, how long will it take for 75% of the NO2(g) to decompose? The decomposition of NO2(g) is second-order in the reactant and the rate constant for this reaction, at 573 K, is 1.1 L/mol s.arrow_forward
- The isomerization of cyclopropane, C3H6, is believed to occur by the mechanism shown in the following equations: C3H6+C3H5k1C3H6+C3H6(Step1)C3H6k2C2=CHCH3(Step2) Here C3H6 is an excited cyclopropane molecule. At low pressure, Step 1 is much slower than Step 2. Derive the rate law for this mechanism at low pressure. Explain.arrow_forwardChlorine dioxide, ClO2, is a reddish-yellow gas that is soluble in water. In basic solution it gives ClO3 and ClO2 ions. 2ClO2(aq)+2OH(aq)ClO3(aq)+ClO2(aq)+H2O To obtain the rate law for this reaction, the following experiments were run and, for each, the initial rate of reaction of ClO2 was determined. Obtain the rate law and the value of the rate constant.arrow_forwardThe following statements relate to the reaction for the formation of HI: H2(g) + I2(g) 2 HI(g)Rate = k[H2][I2] Determine which of the following statements are true. If a statement is false, indicate why it is incorrect. (a) The reaction must occur in a single step. (b) This is a second-order reaction overall. (c) Raising the temperature will cause the value of k to decrease. (d) Raising the temperature lowers the activation energy for this reaction. (e) If the concentrations of both reactants are doubled, the rate will double. (f) Adding a catalyst in the reaction will cause the initial rate to increase.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





