
ORGANIC CHEMISTRY 2-SEMESTER-ACCESS
4th Edition
ISBN: 9781119659532
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 11, Problem 38IP
Interpretation Introduction
Interpretation: To propose a synthesis of
Concept introduction:
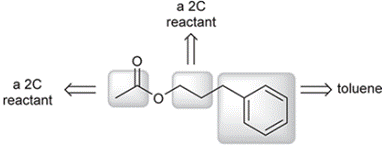
An analysis of the structure of the product suggests the following origins of each of the carbon atoms in the product.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Dimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of
female hamsters, acts as a sexual attractant for the male hamster. Write
an equation for its synthesis from methanethiol.
A synthetic organic molecule, G, which contains both aldehyde and ether functional groups, is subjected to a series of reactions in a multi-step synthesis pathway. In the first step, G undergoes a Wittig reaction, leading to the formation of an alkene, H. Subsequently, H is treated with an ozone (O3) reagent followed by a reducing agent in an ozonolysis reaction, resulting in the formation of two different products, I and J. Considering the functional groups present in G and the nature of the reactions involved, what are the most probable structures or functional groups present in products I and J? A. I contains a carboxylic acid group, and J contains an aldehyde group. B. I contains a ketone group, and J contains an alcohol group. C. I and J both contain aldehyde groups. D. I contains an ester group, and J contains a ketone group. Don't use chat gpt.
A postgraduate student wanted to synthesized two carbonyl compounds known as
compounds M and N using oxidation of alcohol. However, these carbonyl
compounds should have more than 6 carbon atoms.
Draw two (2) possible structural formulae for compounds M and N.
How would you distinguish between compounds M and N?
Chapter 11 Solutions
ORGANIC CHEMISTRY 2-SEMESTER-ACCESS
Ch. 11.1 - Prob. 1CCCh. 11.1 - Prob. 2CCCh. 11.2 - Prob. 1LTSCh. 11.2 - Prob. 3PTSCh. 11.3 - Prob. 2LTSCh. 11.3 - Prob. 6ATSCh. 11.4 - Prob. 3LTSCh. 11.4 - Prob. 7PTSCh. 11.4 - Prob. 8ATSCh. 11.5 - Prob. 4LTS
Ch. 11.5 - Propose an efficient synthesis for each of the...Ch. 11.5 - Prob. 10ATSCh. 11 - Prob. 11PPCh. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Prob. 19PPCh. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Prob. 25PPCh. 11 - Prob. 26ASPCh. 11 - Prob. 27ASPCh. 11 - Prob. 28ASPCh. 11 - Prob. 29ASPCh. 11 - Prob. 30ASPCh. 11 - Prob. 31ASPCh. 11 - Prob. 32ASPCh. 11 - Prob. 33IPCh. 11 - Prob. 34IPCh. 11 - Prob. 35IPCh. 11 - Prob. 36IPCh. 11 - Prob. 37IPCh. 11 - Prob. 38IPCh. 11 - Prob. 39IPCh. 11 - Prob. 42CPCh. 11 - The compound Z-3-hexenyl acetate is one of several...Ch. 11 - When a consumer purchases a tomato, smell is one...Ch. 11 - Prob. 45CPCh. 11 - Prob. 46CP
Knowledge Booster
Similar questions
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forwardSeveral diamines are building blocks for the synthesis of pharmaceuticals and agro-chemicals. Show how both 1,3-propanediamine and 1,4-butanediamine can be prepared from acrylonitrile.arrow_forward
- how to synthesize 2-phenylclohexanone from cyclohexanone?arrow_forwardSynthesize 2-pentanone from 1-butyne. Indicate all the reagents needed.arrow_forwardShow how to convert carboxylic acids to other functional groups, and devisemultistep syntheses using carboxylic acids as starting materials and intermediates.Explain how acid chlorides are used as activated derivatives of carboxylic acidsarrow_forward
- Propose single-step and multistep syntheses of acid derivatives from compoundscontaining other functional groups. Propose multistep syntheses using acid derivatives as starting materials and intermediates.arrow_forwardthe organic compound 2-heptanone, belonging to the ketone family, is responsible for the strong penetrating odor in Roquefort cheeses. Starting from acetylene as the starting reagent, propose a synthesis line with the reaction mechanisms involved for the synthetic obtaining of 2-heptanone and use it as a food additive in analogous cheeses.arrow_forwardReaction of 1-butanol with periodane gives Butanal Propanoic acid 2-Butanone Butanoic acid as the major product.arrow_forward
- Show how to synthesize 3-nitro-4-propoxybenzoic acid from phenol, C6H5OH and any reagents that you need.arrow_forwardShow how to bring about the following conversions using as one step the hydrolysis of a cyano group.arrow_forwardPropose a method for synthesizing 1-bromopentane from 1-pentanolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
