
Concept explainers
Classify each of the following compounds as a normal
a. 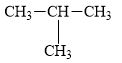
b. 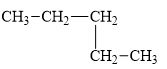
c. 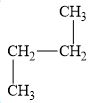
d. 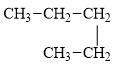
e. 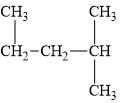
f. 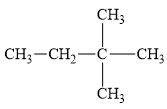
(a)
Interpretation:
The alkane
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound,
Explanation of Solution
The alkane is shown below.
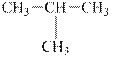
Figure 1
Branched alkanes do not have linear structure and they have alkyl substituents. In the above compound,
The compound,
(b)
Interpretation:
The alkane pentane is to be classified as normal alkane or a branched alkane.
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound, pentane is a normal alkane.
Explanation of Solution
The alkane is shown below.
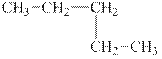
Figure 2
Normal alkanes have linear structure and have no alkyl substituents attached to it. The above compound, pentane has linear structure and has no alkyl substituents. Therefore, it is a normal alkane.
The compound, pentane is a normal alkane.
(c)
Interpretation:
The alkane butane is to be classified as normal alkane or a branched alkane.
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound, butane is a normal alkane.
Explanation of Solution
The alkane is shown below.
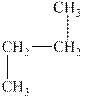
Figure 3
Normal alkanes have linear structure and have no alkyl substituents attached to it. The above compound, butane has linear structure and has no alkyl substituents. Therefore, it is a normal alkane.
The compound, butane is a normal alkane.
(d)
Interpretation:
The alkane pentane is to be classified as normal alkane or a branched alkane.
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound, pentane is a normal alkane.
Explanation of Solution
The alkane is shown below.
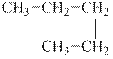
Figure 4
Normal alkanes have linear structure and have no alkyl substituents attached to it. The above compound, pentane has linear structure and has no alkyl substituents. Therefore, it is a normal alkane.
The compound, pentane is a normal alkane.
(e)
Interpretation:
The alkane
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound,
Explanation of Solution
The alkane is shown below.
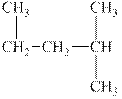
Figure 5
Branched alkanes do not have linear structure and they have alkyl substituents. In above compound,
The compound,
(f)
Interpretation:
The alkane
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound,
Explanation of Solution
The alkane is shown below.
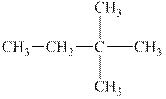
Figure 6
Branched alkanes do not have linear structure and they have alkyl substituents. In the above compound,
The compound,
Want to see more full solutions like this?
Chapter 11 Solutions
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
- The general formula of an alkane is CnH2n+2 . What is the general formula of an (a) alkene? (b) alkyne? (c) alcohol derived from an alkane?arrow_forwardRefer to the given, the structures are: a. cis isomers b. trans isomersc. E isomers d. Z isomerarrow_forwardDraw choral molecules that meet the following description A.chloroalkane. B.an alcoholarrow_forward
- Which of the following does NOT belong to the group? a. alkenes b. alkanes c. alkynes d. unsaturated chains *Choose only one answer and explain.arrow_forwardThe IUPAC name for the following structure will end with....is known as a. ethane b. butane c. hexane d. octanearrow_forwardb. d. Name the following cyclic hydrocarbons: CH, CH, CH-CH,arrow_forward
- 15. What is the parent chain name of the hydrocarbon below? hexane b. hexene a. c. heptane d. heptenearrow_forwardConsider the following isomers of hexene: A. a disubstituted cis- alkene B. the geometric isomer of ISO C. a tri-substituted Z- alkene D. most stable alkene E. has the highest boiling point F.arrow_forwardWrite the condensed formula for the following alcohols and others.a. pentylene glycolb. methyl sec. hexyl carbinolc. ethoxy hexanearrow_forward
- write the formula of thefollowing alkanes:a. heptane b. Nonane c. Octane d. Dodecanee. Undecanearrow_forwardWhich of the following is NOT an isomer of the other three? a. b. с. d.arrow_forwardDraw skeletal structures for the following: a. 5-ethyl-2-methyloctane b. 1,3-dimethylcyclohexane c. 2,3,3,4-tetramethylheptane d. propylcyclopentane e. 2-methyl-4-(1-methylethyl)octane f. 2,6-dimethyl-4-(2-methylpropyl)decanearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





