
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 10.1, Problem 1LTS
Interpretation Introduction
Interpretation:
All the resonance structures of the following radical need to be drawn:
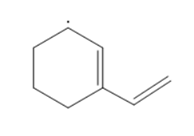
Concept Introduction :
When the bonding cannot be represented by a single Lewis formula, resonance is a means to describe delocalized electrons inside specific molecules or polyatomic ions. With such delocalized electrons, a molecule or ion can be represented by a number of resonance structures.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a stepwise mechanism for the reaction shown below. Use curved arrows to show all bonds broken and formed, draw the structure of all intermediates, and include any formal charges.
I’m supposed to make a resonance structure of this with the same formal charge and I’ve tried at least 7 times with different combinations but I can’t seem to get it right. Can you show me the general direction of where I should go with this? Thanks!
3. For the following structures:
CH3a
C-CH,CH3
C Hy' H
(a) Determine whether each radical is a primary, secondary, or tertiary radical.
(b) Rank these radicals in terms of their relative stability (1 = most stable to 3 = least stable).
4 Draw the
(2) banoggata 1o
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- My book says this compound is not aromatic because it possesses an allene group , which is C=C=C. I dont see where the allene is, can someone show me? I'm confused about how the radical is counted as a C?arrow_forwardH H3C- 1) Using the correct arrow formalism, draw all of the reasonable resonance structures for the radical shown below. 2) Give the structures of A through D: H 1) NaNH2 2-4 2) CH₂CH3 Lindlar's -Br 2 HBr A C D 1) NaNH2 2) -Br Barrow_forwardRe-draw each of the following making sure to include lone pairs and formal charges in each drawingarrow_forward
- Propose the electronic mechanism for the reaction, use curved arrows to show the flow of the electrons ir each step with the respective formal charges.arrow_forwardFirst Writedown which reaction it is? SN1, SN2, E1? Write a stepwise mechanism for the following reactions showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show all necessary lone pairs and formal charges.arrow_forwardDraw the structure of the product in the reaction of aniline with CH,Cl in the presence of AlCl, . Draw applicable formal charges and the appropriate number of hydrogens on the nitrogen atom. Select Draw Rings More Erase : NH2 CH3CI AICI3arrow_forward
- Break compound A into several small (less or equals to 6 carbon atom) molecules. You need to explain the reasons why you choose to break those bonds.arrow_forwardClassify each radical as 1°, 2°, or 3°.arrow_forward-CH=CH-CH2 -CH-Ö: Select all that apply A В Which of the following statements correctly describe resonance for the -CH=CH- -CH-NH-CH3 structures shown? (Select all that apply.) O B has another resonance form because it has an atom bearing a negative charge bonded to an electron-deficient atom. EText Alternative O A has another resonance form because an electron-deficient atom bearing a positive charge is next to a multiple bond. O c does not have another resonance form because it has a localized multiple bond. O D does not have another resonance form because the lone pair on N is held too tightly to be delocalized.arrow_forward
- Draw all significant resonance contributors for the following and label each contributor as major, minor, or contributes equally. CH2 CH CHarrow_forward23. Draw a resonance structure for the radical below and circle the one that is the most stable.arrow_forwardDraw the resulting product(s) from the coupling of the given radicals. Inlcude all applicable electrons and non-zero formal charges. CH3 -CH3 H,C- CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
