
Concept explainers
(a)
Interpretation:
The
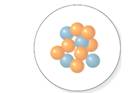
Concept Introduction:
The atomic number is equal to the number of protons of an element whereas sum of number of protons and number of neutrons is equal to mass number.
(b)
Interpretation:
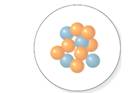
The mass number should be determined for the nucleus which is shown in following diagram.
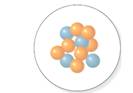
Concept Introduction:
The atomic number is equal to the number of protons of an element whereas sum of number of protons and number of neutrons is equal to mass number.
(c)
Interpretation:
The number of protons should be determined for the nucleus which is shown in following diagram.
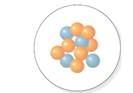
Concept Introduction:
The atomic number is equal to the number of protons of an element whereas sum of number of protons and number of neutrons is equal to mass number.
(d)
Interpretation:
The number of neutrons should be determined for the nucleus which is shown in following diagram.
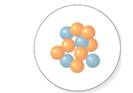
Concept Introduction:
The atomic number is equal to the number of protons of an element whereas sum of number of protons and number of neutrons is equal to mass number.
(e)
Interpretation:
The isotope symbol should be determined for the nucleus which is shown in following diagram.
Concept Introduction:
The atomic number is equal to the number of protons of an element whereas sum of number of protons and number of neutrons is equal to mass number.
A form of chemical element having same atomic number but differ by mass number is known as isotopes. In nuclide notation of isotope, the mass number of the isotope is present in superscript in front of the
The expression is given by:
where, A = mass number and Z = atomic number
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
General, Organic, and Biological Chemistry - 4th edition
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





