
EBK THINKING LIKE AN ENGINEER
4th Edition
ISBN: 8220103633512
Author: OHLAND
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 3RQ
- 3. A phase diagram for carbon and platinum is shown. Assuming the lines shown are linear, we can say the mixture has the following characteristics:
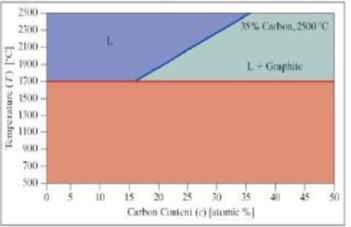
- Below 1700°C, it is a mixture of solid platinum and graphite.
- Above 1700°C, there are two possible phases: a liquid (L) phase and a liquid (L) + graphite phase. The endpoints of the division line between these two phases are labeled on the diagram.
Use the workbook provided to determine the phase of a mixture, given the temperature and carbon content.
| A | B | C | D | E | |
| 1 | |||||
| 2 | |||||
| 3 | Maximum Temperature for Pt + G | 1700 | [oC] | ||
| 4 | |||||
| 5 | |||||
| 6 | |||||
| 7 | Temperature (T) [oC] |
Carbon content (c) [%] |
Temp between L & L+G |
Phase | |
| 8 | |||||
| 9 | 854 | 42 | |||
| 10 | 564 | 20 | |||
- a. Write the equation to describe the temperature of the dividing line between the liquid (L) region and the liquid (L) 1 graphite region in column C. Reference the carbon content found in column B as needed. Add any absolute reference cells you feel are needed to complete this calculation.
- b. Write the conditional statement to determine the phase in column D. For simplicity, call the phases Pt + G, L, and L + G. For points on the line, YOU can decide which phase they are included in.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3. Platinum and gold are completely soluble in both the liquid and solid states. The melting point
of platinum is 3225°F and that of gold is 1945°F. An alloy containing 40% gold starts to solidify
at 2910°F by separating crystals of 15 percent gold in solid. An alloy containing 70% gold starts
to solidify at 2550°F by separating crystals of 37% gold in solid.
Draw the equilibrium diagram to scale on a piece of graph paper and label all points, lines and
areas.
For an alloy containing 70% gold (a) give the temperature of initial and final solidification.
(b) Give the chemical composition and relative amounts of the phases present at 2440°F.
please show work and formulas so that i may learn
the process correctly. if you use a table please
mention which table was used. thank you so
much.
2. Fill in the blank cells of this table:
T( C)
P(kPa)
u(kJ/kg)
Phase description
175
2524.5
325
4000
325
Saturated liquid
Help me please
Chapter 10 Solutions
EBK THINKING LIKE AN ENGINEER
Ch. 10.1 - Type 5 in cell E22 and 13 in cell E23; type =E22 +...Ch. 10.1 - Type 45 into cell G22 and =G22 + 10 in cell H22....Ch. 10.1 - Type 40 into cell A28 and =A28 + 10 in cell D28....Ch. 10.1 - Type 40 into cell A28 and =A28 + 5 in cell G28....Ch. 10.2 - Launch a new worksheet. Type the following Excel...Ch. 10.2 - As part of the design of a high-performance...Ch. 10.3 - Evaluate the following expressions. What is the...Ch. 10.3 - Prob. 8CCCh. 10.4 - This is a continuation of the worksheet you...Ch. 10.5 - Prob. 11CC
Ch. 10.6 - In 1980, the Environmental Protection Agency (EPA)...Ch. 10 - A B C D E F 1 2 45 3 meters 4...Ch. 10 - A B C D E F 1 2 45 3 meters 4...Ch. 10 - A B C D E F 1 2 45 3 meters 4...Ch. 10 - A B C D E F 1 2 45 3 meters 4...Ch. 10 - ICA 10-5 The worksheet shown here was designed to...Ch. 10 - The worksheet provided was designed to calculate...Ch. 10 - Some alternate energy technologies, such as wind...Ch. 10 - The worksheet shown was designed to calculate the...Ch. 10 - The worksheet shown was designed to calculate the...Ch. 10 - Refer to the following worksheet. The following...Ch. 10 - Write the output value that would appear in a cell...Ch. 10 - Write the output value that would appear in a cell...Ch. 10 - Refer to the following worksheet. In all...Ch. 10 - Prob. 14ICACh. 10 - A bioengineer conducts clinical trials on...Ch. 10 - Refer to the Worksheet shown, set up to calculate...Ch. 10 - You are interested in analyzing different implant...Ch. 10 - You have a large stock of several values of...Ch. 10 - We accidentally drop a tomato from the balcony of...Ch. 10 - You are interested in calculating the best place...Ch. 10 - 1. A history major of your acquaintance is...Ch. 10 - Prob. 2RQCh. 10 - 3. A phase diagram for carbon and platinum is...Ch. 10 - 4. A simplified phase diagram for cobalt and...Ch. 10 - 5. You enjoy drinking coffee but are particular...Ch. 10 - 6. In the 1950s, a team at Los Alamos National...Ch. 10 - Use the following phase diagram for questions 7...Ch. 10 - Use the following phase diagram for questions 7...Ch. 10 - Use the following phase diagram for questions 9...Ch. 10 - Use the following phase diagram for questions 9...Ch. 10 - 11. When liquid and vapor coexist in a container...Ch. 10 - 12. The ideal gas law assumes that molecules...Ch. 10 - One of the NAE Grand Challenges for Engineering is...Ch. 10 - 15 Create an Excel worksheet that will allow the...Ch. 10 - Prob. 16RQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Use the following information to answer the next three questions Mobile power plants, such as the one illustrated below, are used in remote locations. Flowing water turns a turbine, which then moves a coil of wire through a magnetic field. Mobile Power Plant Moving water Stream flowing downhill Location I Water turning turbine Location II -Spinning turbine -Permanent magnet Rotating wire coil Split-ring commutator Metal brush 10. The components that connect to the spinning turbine in the illustration on the previous page form a O ammeter O generator O motor 1. O transformer Your reasoning: MacBook Proarrow_forward4. For the A-B alloy the phase diagram below, use the lever rule to find the amount of liquid and solid states and their compositions at Temperature T. Let Cx = 25%B, C =40%B, Cy = 60 %B Complete solubility of two metals in liquid and solid states (Ni -Cu ,Sb - Bi) TA M EM T EMa T Cy A B Concentration of B in A www.substoch com Temperaturearrow_forwardFor Questions 3 and 4: A cup of water was heated to a temperature of 90°. It was placed in a refrigerator that had a temperature of 11°C. The water cooled to a temperature of 17°C in 30 minutes. Let the following variables represent the relevant quantities. L = Temperature of the liquid S= Temperature of the surroundings t = Time B = Initial temperature of the liquid (Temperature at t = 0 k = Cooling constant 3. Using the Equation Solver, find the value of k in the equation L = (B - S)e -kt + S. Remember to set the equation equal to zero and then enter it into the Solver.arrow_forward
- The rod is supported by smooth journal bearings at A, B, and C and is subjected to the two forces F = 310 N and F2 = 540 N . (Figure 1) %3D A F, AF D 0.6 m 0.6 m 0.6 m 0.4 marrow_forwardNewton's Law of cooling states that the rate at which heat is lost by a heated body is proportional to the difference in temperature between the body and the surrounding medium. Match the following statements/phrases with the corresponding item in the drop down box. Choices in the box 1. In accordance with the stated law obove, we have the equation? 2. If a thermometer is taken outdoors where the temperoture is 5 deg C, from a room in which the temperature is 20 deg C and the reading drops 10 deg C in one minute, the working equation is? 3. In the obove problem, separoting the variables and integrating will result to? 4. In the obove problem, how long, in minutes, after its removal from the room will the reading be 6 deg C? 5. In the obove problem, ofter 30 seconds, whot will be the thermometer reading? 5.2 dT(body temp)/dt = k(Temp of medium - 5) dr(body temp)/dt = k(Temp of body - Temp of medium) Temp of body = Cenkt +5 2.5 13.7 dr(body temp)/dt = k(Temp of medium - Temp of body)…arrow_forwardOn an alien planet, the law that expresses the flow of blood through a blood vessel, F, as a function of the radius r of the vessel is f=kr5/2where k is a constant. Certain geological features of this planet produce a gas that changes the radius of blood vessels by 5% at the time of inhalation. What would the corresponding increase in blood flow through the vessel at this time be? Write 1-2 sentences explaining how you found your answer.arrow_forward
- 1- The shape of phase diagram draw by lever rule is * Liquidus-A'bB' B' Liquid |r L+S Solidus - A'dB Solid U' 90 100 В 10 20 30 40 50 60 70 s0 A (o) Composition O True O False Temperaturearrow_forwardRow indexing: Snow fall records stateSnowRecord contains the amount of snow fall (in cm) for various cities. Rows represent a city and columns represent the snow fall for a given month. Assign citySnow Record with all elements of row rowNum. Ex: If stateSnowRecord is [17.3, 20.3; 4.8, 6.2] and rowNum is 2, then citySnowRecord is [4.8, 6.2]. Function > 6 % 7 % 8 9 10 11 12 13 end function citySnowRecord = Get CitySnowFall (stateSnowRecord, rowNum) 2% Get CitySnow Fall: Returns all snow fall values for city in row rowNum % 4 % Inputs: stateSnowRecord matrix of snow fall records for cities for various months rowNum specified row of stateSnowRecord to return stateSnowRecord = [17.3, 20.3; 4.8, 6.2] Outputs: citySnowRecord - all snow fall values for a given city % Assign citySnowRecord with all elements of a row rowNum. citySnowRecord = stateSnowRecord (rowNum, :); Code to call your function > 1 GetCitySnow Fall([17.3, 20.3; 4.8, 6.2], 2) Previous Assessment: 1 of 2 Tests Passed (50%) Save…arrow_forward*29. ssm The drawing refers to one mole of a monatomic ideal gas and shows a process that has four steps, two isobaric (A to B, C to D) and two isochoric (B to C, D to A). Complete the following table by calculating AU, W, and Q (including the algebraic signs) for each of the four steps. B -800.0-K isotherm -400.0-K isotherm 200.0-K isotherm D Volume Pressurearrow_forward
- 1.Based on the water phase diagram below, which of the 2.)A piping system has water operating at 50 °C. Which of the statements below is FALSE: statements below is true for the system to avoid cavitation ок 5ок 100к 150к 200к 250к з00к 350 к 400к 1 TPar 450K 500 K 550K 600 K1 bubbles? XI (hexagonal) X Saturation (or vapor) pressure of water at various temperatures 100 GPa 100 K. 62 GPa VIII VII 10 GPa Saturation Pressure 278 K, 2.1 GPa Temperature T, °C VI V 355.00 K 2.216 GPa 1 GPaXV IX 218 K 620 MPa 248.85 K, 344, 3 MPa 256.164 K, 350.1 MPa 238.5 K. 212.9 MPa t272.99 K. 632.4 MPa Pat, kPa 251.165 K 209.9 MPa -10 0.260 100 MPa Critical poir -5 0.403 Solid Liquid 647 K, 22.064 10 MPa 0.611 5 0.872 1 MPa 10 1.23 15 1.71 XI 100 kPa 20 2.34 (ortho- Freezing polnt at l atm rhombic) Bailing point at 1 atm 373.15 K, 101.325 kPa 273.15 k, 101.325 kPa 25 3.17 10 Pa 30 4.25 40 7.38 12.35 1 kPal 50 Solid/Liquid/Vapour triple point 273.16 К, 611.73 Ра 100 150 101.3 (1 atm) 475.8 100 Pa Vapour 200…arrow_forward1- The shape of phase diagram draw by lever rule is * Liquidus-A'bB' Liquid L+S Solidus-A'dB' Solid 10 20 30 40 50 60 70 80 90 100 A (°o) В Composition True False Temperaturearrow_forwardPlease provide equations that you are using so I can follow along and learn how to do this and include conversions and units. In Jamestown, NY, a coal power plan consumes coal at a rate of 80 tons/hr. When coal burns, it generates approximately 30,000 kJ for every kg of coal burned. The cooling course for the plant is the nearby Chadakoin River nearby and, by code, the power plant cannot reject more than 13.19 x 109 MJ into the river annually. Determine the maximum average power output of this plant and it's maximum thermal efficiency. To check your work, enter the efficiency, as a decimal, in the box provided.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
Mechanical Calculations; Author: Mometrix Academy;https://www.youtube.com/watch?v=FiQw8fpUHMY;License: Standard youtube license