
Concept explainers
Use the following phase diagram for questions 9 and 10.
The following phase diagram is for salt water. There are four possible phases, which depend on the temperature and the sodium chloride content (NaCl).
- Ice and SC = Mixed ice and salt crystals.
- Ice and SW = Ice and salt water.
- SW = Salt water.
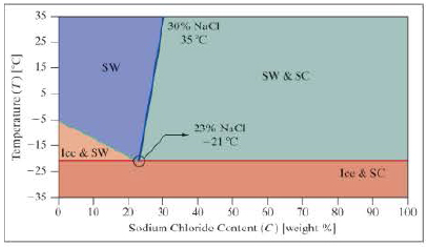
- SW and SC = Salt water and salt crystals.
There are often multiple ways to solve the same problem; here we look at a few alternative ways to determine the phase of the mixture.
- 9. a. In column C, develop the equation for the line dividing the phases of the ice–salt water mix and the salt water. Assume it was written in cell C11 and copied down.
- b. In column D, develop the equation for the line dividing the phases of the salt water and the salt water-salt crystals mix. Assume it was written in cell D11 and copied down.
- c. In column E, write an expression to determine the phase of the mixture.
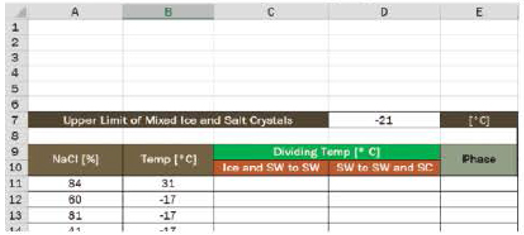
- d. Use conditional formatting to highlight the various phases. Provide a color key.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
Additional Engineering Textbook Solutions
Thermodynamics: An Engineering Approach
Heat and Mass Transfer: Fundamentals and Applications
Vector Mechanics for Engineers: Statics, 11th Edition
Vector Mechanics for Engineers: Statics and Dynamics
Vector Mechanics for Engineers: Statics and Dynamics
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
- I need help matching the following terms to the phrase it describes. Answers are used only once.arrow_forwardHelp me pleasearrow_forwardTask 3 You are an engineer working in a chemical lab, you are conducting an experiment that involve a phase change, your co-worker asked you to explain the relationships between system constants for a perfect gas, answer the following questions to help your colleague to understand the process: Q1. Discuss the difference between gas constant (R) and universal gas constant (Ru)? Explain how the two constants are related? Q2. Propane and methane are commonly used for heating in winter, and the leakage of these fuels, even for short periods, poses a fire danger for residential application. Which gas leakage do you think poses a greater risk for fire? State the reason. Q3. What is the ideal gas equation? Explain the relationship between pressure, temperature and volume in the ideal gas equation? In addition, discuss under what conditions is the ideal-gas assumption suitable for real gases?arrow_forward
- 4. For the A-B alloy the phase diagram below, use the lever rule to find the amount of liquid and solid states and their compositions at Temperature T. Let Cx = 25%B, C =40%B, Cy = 60 %B Complete solubility of two metals in liquid and solid states (Ni -Cu ,Sb - Bi) TA M EM T EMa T Cy A B Concentration of B in A www.substoch com Temperaturearrow_forwardQuestion #5 (Similar to Question #4, You solved, I have attached the question #4)Design a question to calculate the heating load for the building you selected indoor Environment Quality (IEQ) assessment. You will define the types of heat loss (at least two types) in that building (or the floor you are working on or you are familiar with). The indoor temperature is set as 75 F, and you can assume the outdoor temperature based on the location of your building in the winter and any other numbers needed to resolve this question. For all the details as well as the final answer that this question is asking for, clearly indicate the unit for each piece of information.arrow_forward[Engineering Thermodynamics] Please finish the answer with a diagram for reference to me (if needed). Show the answer step by step and clear to see. If you need to answer with words, please type directly. (to avoid cant see clear by handwriting) Thank you. P.S. Use the online software http://www.peacesoftware.de/einigewerte/r134a_e.html or property table and p-h diagram for R134a where necessary.arrow_forward
- please show work and formulas so that i may learn the process correctly. if you use a table please mention which table was used. thank you so much. 2. Fill in the blank cells of this table: T( C) P(kPa) u(kJ/kg) Phase description 175 2524.5 325 4000 325 Saturated liquidarrow_forwardNewton's Law of cooling states that the rate at which heat is lost by a heated body is proportional to the difference in temperature between the body and the surrounding medium. Match the following statements/phrases with the corresponding item in the drop down box. Choices in the box 1. In accordance with the stated law obove, we have the equation? 2. If a thermometer is taken outdoors where the temperoture is 5 deg C, from a room in which the temperature is 20 deg C and the reading drops 10 deg C in one minute, the working equation is? 3. In the obove problem, separoting the variables and integrating will result to? 4. In the obove problem, how long, in minutes, after its removal from the room will the reading be 6 deg C? 5. In the obove problem, ofter 30 seconds, whot will be the thermometer reading? 5.2 dT(body temp)/dt = k(Temp of medium - 5) dr(body temp)/dt = k(Temp of body - Temp of medium) Temp of body = Cenkt +5 2.5 13.7 dr(body temp)/dt = k(Temp of medium - Temp of body)…arrow_forwardI need a literature review about {the Enhancing indoor air quality and safe entertainment in sports halls}......... (introduction about 6 lines) (determination abou 5-6 line) (and 10 reference with link of the reference) with if can photos finally i need the conclusion 5 or 6 line pls urgent.arrow_forward
- Shown in the image is a vertical section of a 66 block grid of a gold mine deposit. Each block is identified with a number and a gold (Au) grade in g/t. As shown, most blocks contain non- valuable or zero ore-grade (G=0). However, the ore-grade of highlighted block #17 is G2= 1.4 g/t, and similarly the ore grades of blocks #28, #39, #50 and #61 are in turn: G3 = 1.2, G4 = 2.3, G5 = 2.8, and G6 = 2.9 g/t. Evaluate the mining methods (o/c or u/g) which should be selected based on economic considerations only.arrow_forwardDirection: Graph the heating curve of ethanol using the information given. Check off each item as you add additional information to your graph so that none is missed. Background information of Ethanol: Boiling Point = 78 0C Melting Point = -114 0C Starting Temperature = -120 0C After two minutes, frozen cold ethanol starts to melt. It takes two minutes to melt completely. After eight more minutes, it begins to boil. It boils for six minutes. Heat is added for two more minutes until ethanol reaches 80 0C. Label “melting” where this takes place. Label “vaporization” where this takes place. Label “phase change” where a phase change occurs. Indicate where ethanol is only a SOLID, only a LIQUID, and only a GAS. Of the three phases, label which phase has: weakest IMF, strongest IMF, and medium IMF.arrow_forwardOU DO: 1) An unknown metal, M, has heated and the following heating curve was created. Answer the following with the letter, letter range (ex: A→B) or a temperature. A) Point(s) where only a gas is present B) Temperature at which vaporization occurs C) Point(s) where a mixture of a solid and a liquid are present D) Point(s) where a gas turns into a liquid E) Temperature at which the substance starts to freeze F) Range where there is only a liquid Temperature (K) 1000 900 800 700 600 500 400 300 200 100 0 -100 -200 Heating curve for Unknown Metal, M C A B D Energy added --> E Farrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





