
Interpreting Data Trans fats are a form of dietary fat that can have significant health consequences. The graph below represents data from a 2004 study that compared the amount of trans fats found in adipose tissue (body fat) of 79 heart attack patients and 167 people who did not have a heart attack. Write a one-sentence summary of the results presented in the graph.
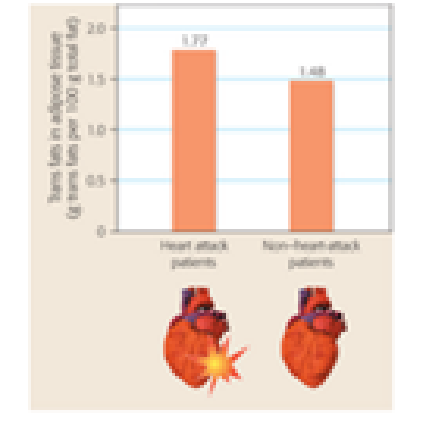
Data from: P. M. Clifton et al., Trans fatty acids in adipose tissue and the food supply are associated with myocardial infarction. Journal of Nutrition 134: 874–879 (2004).
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Campbell Essential Biology with Physiology (5th Edition)
Additional Science Textbook Solutions
Biology Science Notebook
Biology: Concepts and Investigations
Anatomy & Physiology
Campbell Biology (10th Edition)
Human Anatomy & Physiology
Anatomy & Physiology (6th Edition)
- Drink # Glucose Concentration in mM 1 2.455 mM 2 0.307 mM 3 3.750 mM 4 1.100 mM 5 1.500 mM From here, each group needs to convert the concentration of their drink from mM of the diluted drink into g/100mL of the undiluted drink. This is where you come in! The students are stuck at this point and really need your help! Main Goal: Convert the experimental glucose concentration (provided in the table below) from mM (millimolar) to g/100 mL. Hints & Tips Take the 1/100 dilution factor into consideration Molarity will open the doors for you to get to grams. Once at M assume 1L of solution. (Hint: this will help you when converting from M to moles) You will also need to calculate the molar mass of glucose which has the formula C6H12O6 There are 5 steps in total Task 1: Write a step-by-step guide with key words for the Chem 2 students on how to do these calculations. You can use diagrams, a table or just type the steps out. Ensure the steps…arrow_forwardDrink # Glucose Concentration in mM 1 2.455 mM 2 0.307 mM 3 3.750 mM 4 1.100 mM 5 1.500 mM From here, each group needs to convert the concentration of their drink from mM of the diluted drink into g/100mL of the undiluted drink. This is where you come in! The students are stuck at this point and really need your help! Main Goal: Convert the experimental glucose concentration (provided in the table below) from mM (millimolar) to g/100 mL. Hints & Tips Take the 1/100 dilution factor into consideration Molarity will open the doors for you to get to grams. Once at M assume 1L of solution. (Hint: this will help you when converting from M to moles) You will also need to calculate the molar mass of glucose which has the formula C6H12O6 There are 5 steps in total Task 1: Write a step-by-step guide with key words for the Chem 2 students on how to do these calculations. You can use diagrams, a table or just type the steps out. Ensure the steps…arrow_forward. The transition temperatures of the following membranes are -36°C, 23°C, and 41°C. Which membrane correlates with which transition temperature? Explain your answer. Membrane 1) made from entirely phosphatidylcholine and stearate fatty acids Membrane 2) made from entirely phosphatidylcholine and oleate fatty acids Membrane 3) made from entirely phosphatidylcholine and palmitate fatty acidsarrow_forward
- By integrating all the relevant concepts from cellular energetics, explain the aphorism "Fats burn in the flames of carbohydrates” Please answer in at least 5 sentences and include references if necessary. Helping tags: Biology, Chemistry, Biochemistry, Cellular Energeticsarrow_forwardPart A: Explain solubility of vitamins Food additive E307 (alpha-tocopherol, vit. E), and ascorbic acid (vit. C) are essential nutrients in human diet due to their antioxidant properties. They have very different solubilities. In contrast to vit. A, vit. E is insoluble in water. It is recommended that foods rich in vitamin E be taken with fatty ingredients (think salad dressing etc). • Draw the structures of both vitamins and explain their different solubilities in water. • Draw the picture illustrating solvation of vit. C in water and describe intermolecular interactions that facilitate this process. What type of intermolecular interactions help vitamin E be soluble in oils (fatty acids such as linoleic acid)? Part B: Explain observed boiling point The two molecules shown in Fig. 1 are constitutional isomers but have drastically different boiling point. Using a drawing and no more than 5 sentences explain how why compound B (butyramide) has higher boiling point. A O во NH2 153 °C 216…arrow_forwardGiven the graph below, at what pH will the enzyme trypsin perform optimally? Suggest a part in the body where this enzyme would be most probably found to perform best and explain in a max of 5 sentences. Pepsin Trypsin Alkaline phosphatase 3 5 7 9 11 pH Reaction velocity (va)arrow_forward
- The FMO3 enzyme has an important physiological function in people. State the normal substrate of FMO3 in people and describe a genetic polymorphism that can reduce the activity of this enzyme. Describe the consequence to people with the reduced FMO3 activity. Give at least one reference from the primary literature (i.e. a scientific journal) to support your answer.arrow_forwardLingual lipase is an enzyme that is secreted from glands in the oral cavity located near to the tongue (pH=~6.7). This enzyme is specialized to digest triglyceride molecules in the mouth however it is responsible for almost none of the digestion of dietary fat. Food spends only a very brief period of time in the mouth before it is swallowed. The enzyme together with the food then reaches the stomach where the pH is ~2. Considering what you know about tertiary structure, explain using specific examples why this enzyme is not able to participate in lipid digestion in the body.arrow_forwardState Two points to support the statement below, using adequate and sufficient information to clearly explains these points mentioned. Water-soluble vitamins are more biochemically useful than fat-soluble vitamins.arrow_forward
- GenAlex Medical, a leading manufacturer of medical laboratory equipment, is designing a new automated system that can detect normal levels of dissolved glucose (70. to 105 mg/dL), using a blood sample that is as small as 5.0 mL. Calculate the minimum mass in milligrams of glucose that the new system must be able to detect. Be sure your answer has the correct number of significant digits.arrow_forwardHello, please help me with the following 3 questions, thank you so much. Refer to the table and answer the following questions. • Calculate the percentage of the total calories per serving that come from total fat.• Based on a 2,000 calorie diet, what percent of the percent daily value for total fat would be consumed per serving? Calculate the percent if the whole container was consumed.• Based on what you know about trans fats, do you think there are hydrogenated oils in this product? Explain.arrow_forwardTrue or False: a. A polysaccharide formed from beta glycosidic linkages will be used as an energy storage molecule in animals. b. The scientific process can be influenced by peer review, consumer input, and developing technology c. The small molecules that control proteins by allosteric regulation do so binding to the active site.arrow_forward
 Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
