
Chemistry: The Molecular Nature of Matter
7th Edition
ISBN: 9781118516461
Author: Neil D. Jespersen, Alison Hyslop
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 0, Problem 46RQ
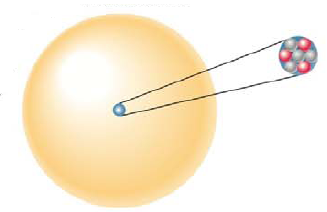
What color are the protons, neutrons, and electrons in the following figure? What element is it? What is the mass number of the atom?
Write the symbol for the atom using the
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 0 Solutions
Chemistry: The Molecular Nature of Matter
Ch. 0 - Practice Exercise 0.1
Using the chapter titles...Ch. 0 - Prob. 2PECh. 0 - Prob. 3PECh. 0 - Prob. 4PECh. 0 - Prob. 5PECh. 0 - Prob. 6PECh. 0 - Prob. 7PECh. 0 - Prob. 8PECh. 0 - Prob. 9PECh. 0 - Practice Exercise 0.10 Aluminum atoms have a mass...
Ch. 0 - Practice Exercise 0.11 How much heavier is the...Ch. 0 - Practice Exercise 0.12 The definition of the size...Ch. 0 - Practice Exercise 0.13 Naturally occurring boron...Ch. 0 - Practice Exercise 0.14
Neon, the gas used in neon...Ch. 0 - Prob. 1RQCh. 0 - 0.2 Knowing what we know today, which of the...Ch. 0 - 0.3 In what ways does the atomic theory affect the...Ch. 0 - Shake a few crystals of salt into your hand and...Ch. 0 - Are there any examples of crystals in nature that...Ch. 0 - 0.6 Heat is one form of energy. If a reaction...Ch. 0 - Can you think of a chemical reaction that starts...Ch. 0 - 0.8 Some large biological molecules rely on...Ch. 0 - 0.9 What elements were formed (a) during the first...Ch. 0 - Prob. 10RQCh. 0 - 0.11 What is the predominant element in the solar...Ch. 0 - Prob. 12RQCh. 0 - What physical factors caused elements and minerals...Ch. 0 - Is this separation still occurring on the earth?Ch. 0 - 0.15 What measuring devices did early chemists...Ch. 0 - 0.16 In your own words, describe how Daltons...Ch. 0 - Which of the laws of chemical combination is used...Ch. 0 - Prob. 18RQCh. 0 - Where in an atom is nearly all of its mass...Ch. 0 - What is a nucleon? Which ones have we studied?Ch. 0 - 0.21 How was the charge-to-mass ratio of the...Ch. 0 - 0.22 How did Robert Millikan determine the charge...Ch. 0 - How was the proton discovered?Ch. 0 - What experiment did Rutherford carry out to...Ch. 0 - Why dont we count the electrons when determining...Ch. 0 - Define the terms atomic number and mass number....Ch. 0 - Prob. 27RQCh. 0 - Write the symbols of the isotopes that contain the...Ch. 0 - What is wrong with the following statement? The...Ch. 0 - 0.30 The atomic number of silver, Ag, is 47 and it...Ch. 0 - A certain element X forms a compound with oxygen...Ch. 0 - Nitrogen reacts with a metal to form a compound in...Ch. 0 - Give the numbers of neutrons, protons, and...Ch. 0 - 0.34 Give the numbers of electrons, protons, and...Ch. 0 - Iodine-131 is used to treat overactive thyroids;...Ch. 0 - Prob. 36RQCh. 0 - 0.37 One chemical substance in natural gas is a...Ch. 0 - 0.38 Carbon tetrachloride contains one carbon and...Ch. 0 - If an atom of carbon-12 had been assigned a...Ch. 0 - 0.40 One atom of has a mass that is 9.0754 times...Ch. 0 - Naturally occurring copper is composed of 69.17%...Ch. 0 - Naturally occurring magnesium (one of the elements...Ch. 0 - N2O is often called nitrous oxide or laughing gas,...Ch. 0 - An element has 24 protons in its nucleus. (a)...Ch. 0 - Iron is composed of four isotopes with the...Ch. 0 - 0.46 What color are the protons, neutrons, and...Ch. 0 - 0.47 Immediately after the big bang, helium and...Ch. 0 - Prob. 48RQCh. 0 - Scientists often validate measurements, such as...Ch. 0 - Sir James Chadwick, discovered the neutron. The...
Additional Science Textbook Solutions
Find more solutions based on key concepts
14.30 For each of the pairs below, predict specific aspects in their NMR spectra that would allow you to disti...
Organic Chemistry
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
For each of the following molecules, (1) draw the correct Lewis structure; (2) label each polar covalent bond w...
General, Organic, and Biological Chemistry (3rd Edition)
In the space provided, draw a mechanism for each of the following transformations:
Organic Chemistry As a Second Language: Second Semester Topics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A hypothetical element X is found to have an atomic weight of 37.45 amu. Element X has only two isotopes, X-37 and X-38. The X-37 isotope has a fractional abundance of 0.7721 and an isotopic mass of 37.24. What is the isotopic mass of the other isotope?arrow_forwardDo the proton and the neutron have exactly the same mass? How do the masses of the proton and neutron compare to the mass of the electron? Which particles make the greatest contribution to the mass of an atom? Which particles make the greatest contribution to the chemical properties of an atom?arrow_forwardGive the complete symbol(ZAX), including atomic number and mass number, of (a) a nickel atom with 31 neutrons, (b) a plutonium atom with 150 neutrons, and (c) a tungsten atom with 110 neutrons.arrow_forward
- Though the common isotope of aluminum has a mass number of 27, isotopes of aluminum have been isolated (or prepared in nuclear reactors) with mass numbers of 24, 25, 26, 28, 29, and 30. How many neutrons are present in each of these isotopes? Why are they all considered aluminum atoms, even though they differ greatly in mass? Write the atomic symbol for each isotope.arrow_forwardWhich of the following is{are) correct? a. 40Ca2 contains 20 protons and 18 electrons. b. Rutherford created the cathode-ray tube and was the founder of the charge-to-mass ratio of an electron. c. An electron is heavier than a proton. d. The nucleus contains protons, neutrons, and electrons.arrow_forward2.90 Naturally occurring europium has an average atomic weight of 151.964 amu. If the only isotopes of europium present are 151Eu and 153Eu, describe how you would determine the relative abundance of the two isotopes. Include in your description any information that would need to be looked up.arrow_forward
- What are the different kinds of particles in the atoms nucleus? Compare their properties with each other and with those of an electron.arrow_forwardThe isotope of an unknown element, X, has a mass number of 79. The most stable ion of this isotope has 36 electrons and has a 2 charge. Which of the following statements is(are) true? For the false statements, correct them. a. This ion has more electrons than protons in the nucleus. b. The isotope of X contains 38 protons. c. The isotope of X contains 41 neutrons. d. The identity of X is strontium, Sr.arrow_forwardCompounds of europium. Eu, are used to make color television screens. The europium nucleus has a charge of +63. How many electrons are there in the neutral atom? in the Eu3+ ion?arrow_forward
- Xenon An isotope of xenon has an atomic number of 54 and contains 77 neutrons. What is the xenon isotope’s mass number?arrow_forwardHow many protons and neutrons are contained in the nucleus of each of the following atoms? Assuming each atom is uncharged, how many electrons are present? a.56130Ba c .2246Ti e.36Li b.56136Ba d.2248Ti f.37Liarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
The Bohr Model of the atom and Atomic Emission Spectra: Atomic Structure tutorial | Crash Chemistry; Author: Crash Chemistry Academy;https://www.youtube.com/watch?v=apuWi_Fbtys;License: Standard YouTube License, CC-BY