
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN: 9781305580343
Author: Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
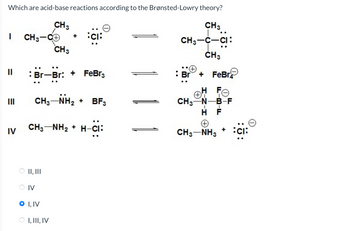
Transcribed Image Text:Which are acid-base reactions according to the Brønsted-Lowry theory?
CH 3
I
CH3-C+
CH3
Br-Br: + FeBг3
IV
CH 3-NH2 + BF3
H-CI:
CH3NH2+H-CI
CH3
CH3-C-CI:
CH3
Br + FeBr
H FO
CH3-N-B-F
H F
A
CH3-NH3
II, III
IV
O I, IV
I, III, IV
SAVE
AI-Generated Solution
info
AI-generated content may present inaccurate or offensive content that does not represent bartleby’s views.
Unlock instant AI solutions
Tap the button
to generate a solution
to generate a solution
Click the button to generate
a solution
a solution
Knowledge Booster
Similar questions
- Write equations to illustrate the acid-base reaction when each of the following pairs of Brnsted acids and bases are combined: Acid Base a.HOCl H2O b.HClO4 NH3 c.H2O NH2 d.H2O OCl e.HC2O4 H2Oarrow_forwardArsenic acid (H3AsO4) is a moderately weak triprotic acid. Write equations showing its stepwise dissociation. Which of the three anions formed in these reactions will be the strongest Brnsted base? Which will be the weakest Brnsted base? Explain your answers.arrow_forwardIn each of the following acid-base reactions, identify the Brnsted acid and base on the left and their conjugate partners on the right. (a) C2H5N(aq) + CH3CO2H(aq) C5H5NH+(aq) + CH3CO2(aq) (b) N2H4(aq) + HSO4(aq) N2H5+(aq) + SO42(aq) (c) [Al(H2O)6]3+ (aq) + OH(aq) [Al(H2O)5OH]2+ (aq) + H2O+()arrow_forward
- Explain the difference between a strong acid and a weak acid.arrow_forwardIn each of the following acid-base reactions, identify the Brnsted acid and base on the left and their conjugate partners on the right. (a) HCO2H(aq) + H2O() HCO2(aq) + H3O+(aq) (b) NH3(aq) + H2S(aq) NH4+(aq) + HS(aq) (c) HSO4(aq) + OH(aq) SO42(aq) + H2O+()arrow_forwardClassify each of the following as a strong acid, weak acid, strong base, or weak base in aqueous solution. a. HNO2 b. HNO3 c. CH3NH2 d. NaOH e. NH3 f. HF g. h. Ca(OH)2 i. H2SO4arrow_forward
- Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced equations for the reactions causing the solution to be acidic or basic. The relevant Ka, and Kb values are found in Tables 13-2 and 13-3. a. Sr(NO3)2 b. NH4C2H3O2 c. CH3NH3Cl d. C6H5NH3ClO2 e. NH4F f. CH3NH3CNarrow_forwardFor oxyacids, how does acid strength depend on a. the strength of the bond to the acidic hydrogen atom? b. the electronegativity of the element bonded to the oxygen atom that bears the acidic hydrogen? c. the number of oxygen atoms? How does the strength of a conjugate base depend on these factors? What type of solution forms when a nonmetal oxide dissolves in water? Give an example of such an oxide. What type of solution forms when a metal oxide dissolves in water? Give an example of such an oxide.arrow_forwardWhich of the following substances are acids in terms of the Arrhenius concept? Which are bases? Show the acid or base character by using chemical equations. a P4O10 b Na2O c N2H4 d H2Tearrow_forward
- Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced chemical equations for the reactions causing the solution to be acidic or basic. The relevant Ka and Kb values are found in Tables 13-2 and 13-3. a. NaNO3 b. NaNO2 c. C5H5NHClO4 d. NH4NO2 e. KOCl f. NH4OClarrow_forwardClassify each of the following statements as true or false: aAll Brnsted-Lowry acids are Arrhenius acids. bAll Arrhenius bases are Brnsted-Lowry bases, but not all Brnsted-Lowry bases are Arrhenius bases. c HCO3 is capable of being amphoteric. d HS is the conjugate base of S2. eIf the species on the right side of an ionization equilibrium are present in greater abundance than those on the left, the equilibrium is favored in the forward direction. f NH4+ cannot act as a Lewis base. gWeak bases have a weak attraction for protons. hThe stronger acid and the stronger base are always on the same side of a proton transfer reaction equation. iA proton transfer reaction is always favored in the direction that yields the stronger acid. jA solution with pH=9 is more acidic than one with pH=4. kA solution with pH=3 is twice as acidic as one with pH=6. lA pOH of 4.65 expresses the hydroxide ion concentration of a solution in three significant figures.arrow_forwardFor which of the following solutions must we consider the ionization of water when calculating the pH or pOH? (a) 3108M HNO3. (b) 0.10 g HCl in 1.0 L of solution. (c) 0.00080 g NaOH in 0.50 L of solution. (d) 1107M Ca(OH)2. (e) 0.0245 M KNO3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co