
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
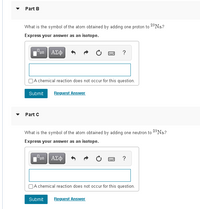
Transcribed Image Text:Part B
What is the symbol of the atom obtained by adding one proton to 2*Na.?
Express your answer as an isotope.
ΑΣφ
OA chemical reaction does not occur for this question.
Submit
Request Answer
Part C
What is the symbol of the atom obtained by adding one neutron to 28N.?
Express your answer as an isotope.
ΑΣφ
?
A chemical reaction does not occur for this question.
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- An unknown element is determined to have two naturally occurring isotopes. Isotope 1 is 68.11% abundant and has a mass of 68.925580 amu, and isotope 2 has a mass of 70.9247005 amu. Determine the atomic mass of the unknown element. Express your answer to four significant figures.arrow_forwardUranium-238 undergoes radioactive decay until a stable isotope is reached. One of the steps is shown below. The chief would like to know what new elements are being produced. Determine the atomic mass, atomic number, and symbol of the new element based on the equation below. 4 227 Ac → ? + ½ ½ a >? 2 89 Atomic Mass Atomic Number Symbol Karrow_forwardBoron has two stable isotopes. What is the approximate ratio of abundance: 10B ___ 11B? 1:5, 1:3, 1:4arrow_forward
- Explain your understanding: Use your own words and captured images from the simulation to show you can: Define “isotope” using mass number, atomic number, number of protons, neutrons and electrons. Compare and contrast: element, atom, isotope. Given the number of protons, neutrons and electrons, find the mass and name of an isotope. Given the name of an element and the number of neutrons, find the mass of an isotope.arrow_forward1- For each of the following atoms, calculate the number of protons and neutrons in the nucleus and the number of electrons outside the nucleus (assume neutral atoms). element number of number of number of electrons mass number (A) symbol 126C protons (Z) neutrons 12 10 12 10 14 27 10 23,1Na 11 17 18 2- Calculate the atomic mass to four significant figures for carbon, given the following data: Isotope Exact Atomic Mass (amu) 12.00000 13.00335 Abundance in Nature (%) 98.89 1.110 12C 13Carrow_forwardThe proton and the electron have about the same mass. Most of the mass of an atom is due to The neutron is electrically neutral. the protons and neutrons. The mass number is the number of The charge of a proton is equal, but opposite, to the charge of a neutron. protons. True False More information needed to make a determinationarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY