
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN: 9781938168390
Author: Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher: OpenStax
expand_more
expand_more
format_list_bulleted
Question
Please correct answer and don't use hand rating
Transcribed Image Text:+
Question 4 of 10
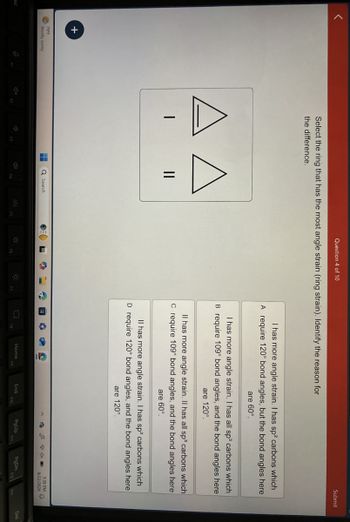
Select the ring that has the most angle strain (ring strain). Identify the reason for
the difference.
Submit
A A
||
I has more angle strain. I has sp² carbons which
A require 120° bond angles, but the bond angles here
are 60°.
I has more angle strain. I has all sp³ carbons which
B require 109° bond angles, and the bond angles here
are 120°.
Il has more angle strain. Il has all sp³ carbons which
C require 109° bond angles, and the bond angles here
are 60°.
Il has more angle strain. I has sp² carbons which
D require 120° bond angles, and the bond angles here
are 120°.
79°F
Mostly sunny
Search
Home
End
PgUp
PgDn
F10
F11
5:39 PM
9/22/2024
Del
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Pleaseeee helpppparrow_forwardWhich of the following compounds is a valid Lewis structure of a hydrocarbon? H H-C-Ö-H H HH H-C-N-H H I II H H-C-C-H H III HH H-C-C-H I НН IV I, II, and IV are valid structures of hydrocarbons. Only III is a valid structure of hydrocarbons. All of these compounds are valid structures of hydrocarbons. Only IV is a valid structure of hydrocarbons. III and IV are valid structures of hydrocarbons.arrow_forwardwhat class is this compound?arrow_forward
- Rank the indicated C-C bonds in increasing order of bond length. O | < || < |||< IV O IIarrow_forwardLine-bond structures appear to imply that there are two different isomers of 1,2-dibromobenzene, one with the bromine-bearing carbon atoms joined by a double bond and one with the bromine-bearing carbons joined by a single bond. In fact, though, there is only one 1,2-dibromobenzene. Explain. H H. C. Br H. .C. Br || || .C and 1,2-Dibromobenzene Br Brarrow_forwardSafari File Edit View History Bookmarks Window Help 36% O A АBС Thu 3:08 PM 5. The general formula of alkene is C„H2n. (a) The relative molecular mass of an alkene X is 70.0 . Find out the molecular formula of X. (Relative atomic masses: C = 12.0, H = 1.0) (b) X is a straight chain alkene showing cis-trans isomerism. (i) Give the structural formula of X. (ii) Draw a 3-dimension diagram of the trans-isomer of X.arrow_forwardWhich of the following compounds is not an alkane? A or B or Carrow_forwardA BE M (c) W Chapter... H₂N Type of hydrocarbon: O alkane alkene alkyne arene X Type of structure: straight-chain branch cyclic aromatic X 73,181 4 с tv G ● JUL 26 Garrow_forwardWhy is the C-C bond length of an alkyne shorter than the C-C bond length of an alkene? O The orbitals are physically smaller due to mores character. O Pi bonds are weaker so more pi bonds creates a shorter bond. O There's a triple bond instead of a double bond.arrow_forwardPlease draw an alkane that has at least one primary, one secondary, and one tertiary carbon. Then take molecule and react it with F^2 (Identify all mono halogenated products for each reaction)arrow_forwardWhich structures represent R-3-methylhexane? H III, IV I, ||| II, IV I, II || Σilliy III IV Iarrow_forwardNaming branched alkanes Name the following organic compounds: compound CH₁₂ | - - CH₂ — CH₂▬ CH₂ | CH - name CH3 血 - - - CH2 CH2 CH2 - - CH3 CH2 — CH₂ — C - - - - CH₂ — CH₂ — CH3 CH, CH₂ CH3 - CH | CH₂ - CH2- - П CH2-CH3 ☐arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning