
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
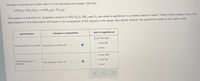
Transcribed Image Text:Nitrogen monoxide and water react to form ammonia and oxygen, like this:
4 NO(g)+6H,0(g) → 4 NH3(g)+502(9)
The reaction is endothermic. Suppose a mixture of NO, H,0, NH, and O, has come to equilibrium in a closed reaction vessel. Predict what change, if any, the
perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left.
perturbation
change in composition
shift in equilibrium
O to the right
O to the left
The temperature is raised. The pressure of NH3 will
(none)
O to the right
O to the left
The temperature is
lowered.
The pressure of NO will
?
O (none)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- please see attached imagearrow_forwardSulfur dioxide and oxygen react to form sulfur trioxide, like this: 2 SO2(g)+O2(9) 2 SO3(g) The reaction is exothermic. Suppose a mixture of SO,, O, and SO, has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium O to the right O to the left: The temperature is raised. The pressure of 0, will O (none) to the right O to the left The temperature is lowered. The pressure of SO3 will 1? O (none) Submit A Continue MacBook Air O O Oarrow_forwardHydrogen sulfide and oxygen react to form sulfur dioxide and water, Ilike this: 2H,S(g)+30,(g) → 2so,(g)+2H,0(g) Suppose a mixture of H,S, O2, SO, and H,O has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium to the right The pressure of H,S will v ? to the left Some So, is removed. go up. go down. not change. The pressure of O, will (none) to the right The pressure of H,S will ? Some 0, is added. to the left The pressure of SO2 will (none)arrow_forward
- Danie Carbon dioxide and water react to form methane and oxygen, like this: CO2(9)+2H,O(9) → CHĄ(g)+2O¿(g) The reaction is endothermic. Suppose a mixture of CO,, H,O, CH, and O, has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium O to the right The temperature is raised. The pressure of H20 will ? O to the left O (none) O to the right The temperature is lowered. O to the left The pressure of CH4 will ? O (none) Submit Assi Continue O 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use I Privacy Center| Ac 43,203 FEB 18 PP ESS MacBook Airarrow_forwardHydrogen sulfide and oxygen react to form sulfur dioxide and water, like this: 2H,s(g)+30,(g) → 2So,(g)+2H,0(g) Suppose a mixture of H,S, 0,, SO, and H,0 has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium O to the right The pressure of 0, will ? Some H,S is removed. O to the left The pressure of SO2 will ? O (none) to the right The pressure of H,S will ? Some H,0 is added. to the left The pressure of O, will ? O (none)arrow_forwardNitrogen and hydrogen react to form ammonia, like this: N,(9)+3H,(g) → 2 NH3(g) The reaction is exothermic. Suppose a mixture of N,, H, and NH, has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. olo perturbation change in composition shift in equilibrium Ar to the right The temperature is lowered. to the left The pressure of N2 will (none) to the right to the left The temperature is raised. The pressure of NH3 will v ? go up. (none) go down. not change. ?arrow_forward
- Ammonium carbamate, NH4CO₂NH₂, decomposes as follows: NH,CO,NH,(s)→ 2NH,(g) + CO, (g) Starting with only the solid, it is found that at 40.0 °C the total gas pressure (NH3 and CO₂) is 0.463 atm. Calculate the equilibrium constant Kp. Round your answer to 3 significant digits. 31 K₂ = 11 Kp x10 X Śarrow_forwardThe equilibrium constant, Kc, for the following reaction is 83.3 at 500 K. PCI3(g) + Cl₂(g) =PC15(9) Calculate the equilibrium concentrations of reactant and products when 0.451 moles of PCI3 and 0.451 moles of Cl₂ are introduced into a 1.00 L vessel at 500 K. [PCI3] = [Cl₂] [PCI5] = = ΣΣΣarrow_forwardHydrogen sulfide and oxygen react to form sulfur dioxide and water, like this: 2H25(g)+302(g) - 2502(g)+2H2O(g) Suppose a mixture of H2S, O2, SO2 and H2O has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium to the right The pressure of H₂S will ? to the left Some H₂O is added. The pressure of O2 will ? ○ (none) to the right The pressure of H₂S will ? Some O2 is removed. to the left The pressure of SO2 will ? ○ (none)arrow_forward
- Carbon disulfide and oxygen react to form carbon dioxide and sulfur dioxide, like this: cs,(e)+30,(2) → CO,(2)+2S0,(g) Suppose a mixture of CS,, O,, CO, and SO, has come to equilibrium in a closed reaction vessel. Predict what change, if any, the pertur below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium Oto the right The pressure of O, will Some CS, is removed. O to the left The pressure of CO, will O (none) O to the right Some Co, is added. The pressure of CS, will O to the left The pressure of O, will O (none) Explanation Check P Type here to search O 2022 McGraw H LLC Al Rights Reserved Terms of Use Pivac 99+ ook Alt Ararrow_forwardMethane and water react to form hydrogen and carbon monoxide, like this: CH₁(9)+H₂O(g) → 3H2(g)+CO(g) The reaction is endothermic. Suppose a mixture of CH 4, H2O, H2 and CO has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition shift in equilibrium to the right to the left The temperature is raised. The pressure of CH4 will ? ○ (none) to the right to the left The temperature is lowered. The pressure of CO will ? O (none) ×arrow_forwardHydrogen chloride and oxygen react to form water and chlorine, like this: 4HCI(g)+O,(g) → 2H,0(g)+2Cl,(g) Suppose a mixture of HCI, 0,, H,0 and Cl, has come to equilibrium in a closed reaction vessel. Predict what change, if any, the perturbations in the table below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. olo perturbation change in composition shift in equilibrium Ar O to the right The pressure of O, will ? to the left Some HCl is removed. The pressure of H20 will (none) to the right The pressure of HCl will ? Some H,0 is added. to the left The pressure of O, will (none)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY