
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN: 9781305580343
Author: Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Please show the mechanism with appropriate arrow formalism.
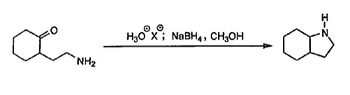
Transcribed Image Text:NH₂
H3O X NaBH4, CH3OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Similar questions
- ||| A ALEKS-Katelyn Pearson... 1 Apromatincat Rom Fecr] ATOMS, IONS AND MOLECULES Predicting the formula of ionic compounds with common... Write the empirical formula for at least four ionic compounds that could be formed from the following ions: OH, Fe²+, 103, NH b Answered. The following X O Garrow_forwardLiN(-Pr)2 -78°С, THF; NaH 25 °С, THF Br 'Br b. H3O* heat MeO OMe С. NaOEt ELOH, heat d. Br2 Brz (еxcess) cat. ACOH NaOH, H2Oarrow_forwardDescribe the steps you would take to write the chemical formula for the compound bromine pentafluoride. Answer in complete sentences. Make sure to use the formula editor when you are writing subscripts. I !!! H Normal А А Enter your answer here ||| BIUS √x 20 Txarrow_forward
- Need help from A-Darrow_forwardThe chemical structure of lactic acid (C,H,O,) is shown below. Highlight each atom that is in a hydroxy group. H :0 : ? Н —О С — С О — Н Н —С —Н H :0 :arrow_forwardFluorine is an example of a(n) O molecular compound O ionic compound O atomic element molecular element a 立arrow_forward
- Some salts make hydrates when there is moisture around. Some hydrate samples can absorb lights in visible legion. Therefore, they show some colors. Thus, hydrates can be used to detect the moisture in the environment. Let's say you are working in a laboratory with a group and you are the only student who has taken chemistry courses. Your laboratory received a sample of cobalt(II) chloride which has the formula CoCl2·xH2O. Let's say your boss asked you to find the formula of this hydrate salt sample since you are the only chemist there. From your laboratory experience, simply explain the experimental procedure you would follow to find the formula of this unknown sample. Then, use the given data for the calculation part. Mass of crucible: 32.27g Mass of crucible + unknown hydrate: 33.92 g Color of unknown hydrate: purple Mass of crucible + anhydrous form of hydrate: 33.41 g Color of anhydrous form: sky bluearrow_forwardThe lattice energy of CsF is -744 kJ/mol, whereas that of BaO is -3029 kJ/mol. Explain this large difference in lattice energy.arrow_forwardImagine that you are given 0.2500 g of a sample of copper(II) sulfate pentahydrate (CuSO4 • 5 H2O). You very carefully heat the compound for an extended period of time to drive off water, after which you determine the mass of the remaining solid to be 0.1598 g. Determine whether the data given confirm the formula of the hydrate. You must show any relevant calculationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning