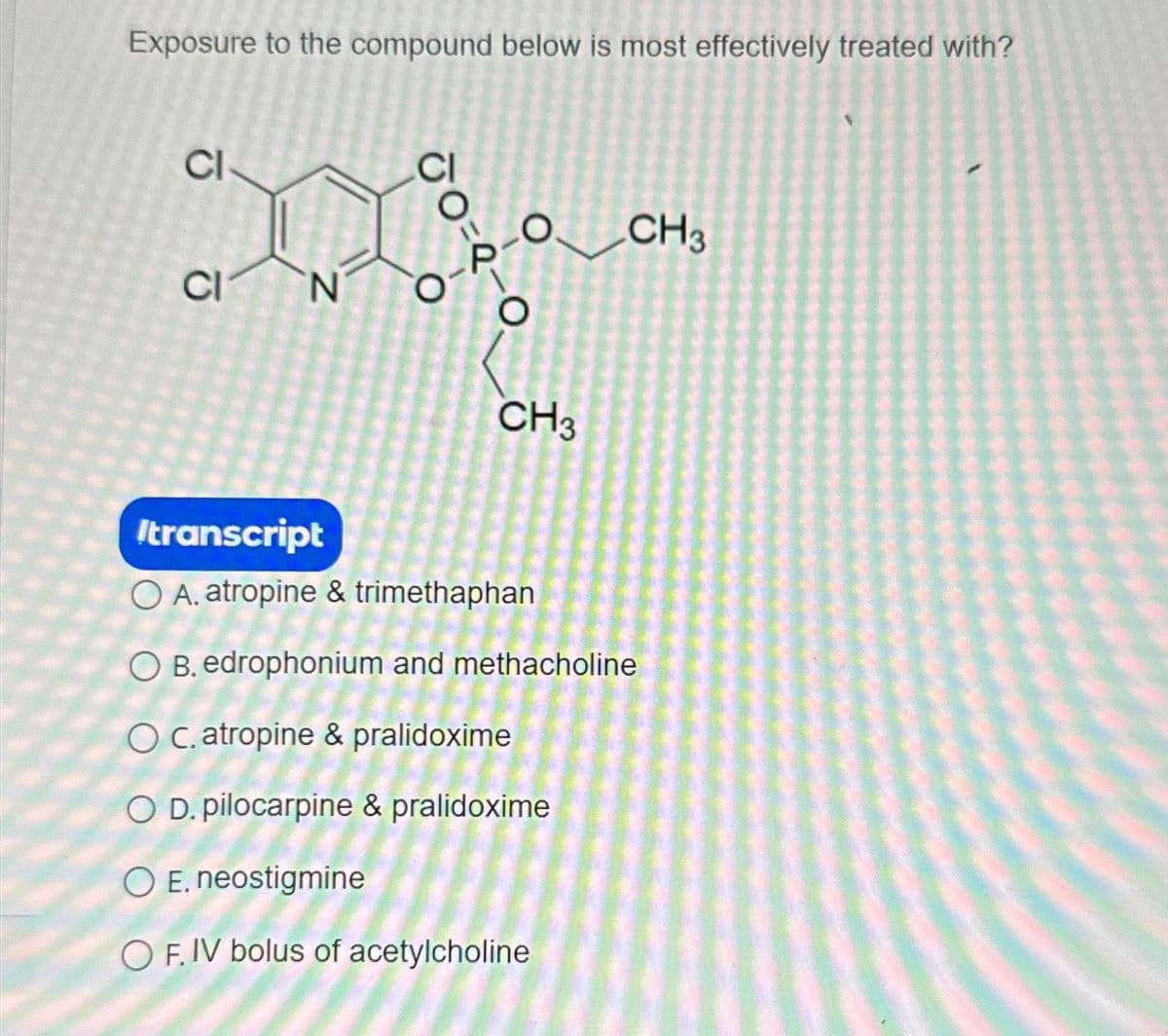
Exposure to the compound below is most effectively treated with? CI CI N CI O O CH3 CH3
Q: Which of the following is TRUE? Chylomicrons have more triglycerides than HDLs. HDL carries both…
A: In the human body, lipoproteins are essential for lipid transport and metabolism. The equilibrium of…
Q: 1. 2. 3. 4. 5. 6. The Ramachandran plot indicates: The preferred conformations about C1-C bond The…
A: phi (Φ) and psi (ψ) angles are known as dihedral angles. They dictate the peptide conformation. phi…
Q: Which of the following sites represents the anomeric carbon? О ОН B O с A E D C A D CH3OH ОН ОН E OH
A: Carbohydrates are poly hydroxy aldehydes or ketones.Carbohydrates can be classified as…
Q: Choose the structure of phosphatidylcholine from those shown below ON< R₁ R₁ R2 R₂ X NH3 OH A B I
A: There are four classes of biological macromolecules- nucleic acid, proteins, lipids and…
Q: 1. The liver produces glucose to the rest of the body between meals by breaking down glycogen stores…
A: Glucose is the body’s principle source of energy that is derived as a macronutrient in certain…
Q: Synthesis of the activated form of acetate (acetyl-CoA) is carried out in an ATP-dependent process.…
A: The nucleotide coenzyme adenosine triphosphate (ATP) is the most important form of chemical energy…
Q: Which amino acid residue would be most effective at forming a strong intermolecular interaction with…
A: Amino acids have chemically diverse side groups that can participate is different types of…
Q: Emanuel's body metabolizes caffeine at a rate of 14.4% per hour (so the amount of caffeine in…
A: Caffeine is a naturally occurring central nervous system (CNS) stimulant. Caffeine is metabolized…
Q: Write equations for the hydrolysis of ATP and ADP. Use abbreviations such as ATP for adenosine…
A: ATP (adenosine triphosphate) functions as the main source of energy for cells. Energy is transferred…
Q: Consider a uniport system where a carrier protein transports an uncharged substance A across a cell…
A: The following equation describes the mathematical relation for the change in free energy () SHAPE…
Q: Which process is most responsible for the sickling of cells seen in sickle cell disease? A)…
A: Sickle cell disease, also known as sickle cell anemia, is a genetic blood disorder characterized by…
Q: How are oxytocin and vasopressin related?!. They are synthesized in the hypothalamus.II. They have 8…
A: Oxytocin is a neuropeptid. that is released by the posterior pitutary gland. This hormone has been…
Q: results from hydrogen bonding between the backbone constituents of the overall shape of a…
A: There are four classes of biological macromolecules; nucleic acid, proteins, lipids and…
Q: Indicate how the observed Km of an enzyme would change in the presence of inhibitors having the…
A: The Km is known as Michaelis constant, it is the concentration of substrate at which reaction…
Q: Single stranded DNA.
A: DNA:Its full form is deoxyribonucleic acid.It carries genetic instructions for the development,…
Q: Which of the following are roles of the distal histidine in hemoglobin? to help to keep the heme in…
A: The distal histidine in hemoglobin plays a crucial role in its functioning. Let's analyze the…
Q: hemoglobin.
A: Hemoglobin, a crucial protein found in red blood cells, plays a pivotal role in the efficient…
Q: Consider a small protein containing 98 amino acid residues. The protein backbone will have 194 bonds…
A: Assuming that each of the 194 rotatable links in the protein backbone can have three alternative…
Q: Choose the TRUE statement. A phospholipid micelle (not a liposome) can be characterized as Forming…
A: Phospholipids are important components of cell membranes and can form various structures in aqueous…
Q: All of the following statements about glucose are true except: Glucose is metabolized in the…
A: Glucose is a simple sugar (monosaccharide) that serves as a primary source of energy for living…
Q: The F₁F. - ATP synthase is a molecular machine that converts the proton motive force into enzymatic…
A: The molecular device known as F1F0-ATP synthase transforms the proton motive force into enzymatic…
Q: The monosaccharides shown below are H но-н H- OH \\ HOH но-н но-н H-OH HO H H-OH CH₂OH Enantiomers…
A: Isomers are defined as the two or more molecules with the same molecular formula but different…
Q: A mutation of Lysine to Alanine in the middle of TM1 has been associated with increased GPCR…
A: The lipid bilayer is a thin polar membrane. It is made up of two layers of lipid molecules. The…
Q: What is the role of FAD in the preparation of pyruvate to enter the citric acid cycle? to perform a…
A: The question is asking about the role of Flavin adenine dinucleotide (FAD) in the process of…
Q: In one type of hemoglobin variant, lysine EF6 is replaced by an aspartic acid residue. What would be…
A: The replacement of lysine (K) by aspartic acid (D) at position EF6 in hemoglobin affects the…
Q: How can an enzyme produce a chiral product from a nonchiral or prochiral substrate?
A: The objective of this question is to understand how an enzyme can produce a chiral product from a…
Q: Part A-Carbon dioxide transport The diagram below depicts the transport of carbon dioxide in blood,…
A: Carbon dioxide molecules are transported in the blood from body tissues to the lungs. Several…
Q: What is the minimal resolution required for gleaning insights into structures of drug-drug target…
A: The minimal resolution required for gleaning insights into the structures of drug-drug target…
Q: Representations of sequencing chromatograms for variants of the a chain of human hemoglobin are…
A: The sequencig chromatograms given in question are generated as we conduct Automated DNA sequencing…
Q: How many oxygen atoms are reduced from each nadh and fadh
A: By the end of citric acid cycle most of the energy has been converted into high energy electrons in…
Q: The second messenger cyclic AMP (CAMP) is synthesized from ATP by the activity of the enzyme…
A: Some signalling molecules cannot diffuse across the biological membranes so they communicate their…
Q: How many different reactions (enzymes) in the glycolytic pathway (steps 1 - 10) involve ATP either…
A: In glycolysis, a 6-carbon molecule of glucose-6-phosphate is broken down into 2 molecules of…
Q: The Ki for a competitive inhibitor pyrrole-2-carboxylate is 0.05M. Calculate the initial velocity if…
A: A form of enzyme inhibitor known as a competitive inhibitor pits the substrate against the enzyme's…
Q: You are a genetic analyst and you have a patient with inherited defects of glycolysis. Specifically,…
A: Hexokinase is an enzyme that catalyzes the rate-limiting first step of glycolysis.The…
Q: Which of the following stretches of RNA is most likely to form a hairpin with 4 bases making up the…
A: A nucleotide (pentose sugar + nitrogenous base + phosphate group) is a phosphoric acid ester of a…
Q: Which of the following statements about 2,3-bisphosphoglycerate (2,3-BPG) are TRUE. 2,3-BPG lowers…
A: Let's assess each statement to determine which ones are true:1. 2,3-BPG lowers the affinity of…
Q: 6. List the main similarities and differences between transposons, retrotransposons, group II…
A: Retroviruses are RNA viruses that use reverse transcriptase to convert their RNA genome into DNA…
Q: Using the values of pKas in the table below, calculate the pỈ for DTLH. Properties of some amino…
A:
Q: Frog muscle cells and the solution bathing the cells contain ions at different concentrations. The…
A: The concentrations of Na+, Ca2+, K+, and Cl- are shown in the following table. Temperature = 25…
Q: polysaccharides
A: Polysaccharides are part of the complex carbohydrate category, forming from multiple monosaccharide…
Q: Lipids in a bilayer can diffuse laterally at a relatively fast rate, but "flip-flop" from one…
A: Flippases, floppases, and scramblases are phospholipid translocators present in the membrane…
Q: 2 T/F High intracellular concentrations of AMP would increase the ATP-synthesis pathways A mutation…
A: ATP is the currency the cell spends on metabolic transactions. Acetyl CoA is one of the substrates…
Q: Proline racemase catalyzes the conversion between L-proline and D-proline. The Km and kcat for this…
A: Proline racemase is an enzyme that catalyzes the racemization of L-proline (S configuration) to…
Q: Mutations of Lysine to Alanine residues near the membrane water interface disrupts binding to the…
A: Lipids are a chemically diverse group with two common characteristics: low solubility in water and…
Q: 1. Start with the Michaelis-Menten equation and convert it to a double-reciprocal equation. Show how…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: Sarin is an inhibitor of acetylcholinesterase. Draw a mechanism that shows this.
A: Before going into the mechanism by which sarin inhibits acetylcholinesterase, we need to know a bit…
Q: Adenosine triphosphate (ATP) is the main energy currency used in cells. ATP hydrolysis is coupled…
A: The structure of ATP comprises of a nucleoside triphosphate, consisting of a nitrogenous base…
Q: 1) The approximate length of a DNA double helix is around 0.34nm per base pair. If the extracted D…
A: 1) A somatic human cell has 23 pairs of chromosomes, that is 46 pieces of DNA. The combined length…
Q: What are the benefits of measuring the initial rate of a reaction V. for use in kinetic studies?…
A: Recall that the rate of a chemical reaction is dC/dt where dC is the change in the analyte…
Q: Equation 7a of the text, (max/KM1) [S]-(v'max/KM2) [P] 1+ [S]/KM1 + [P]/KM2 gives the expression for…
A: The following are given in the equation:V represents the rate of the reaction.S represents the…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Which choice properly identifies how racemic epinephrine is prepared? A) Bovine epinephrine prepared for vetrinary use B) A mixture of 50% each of dextrorotery and levorotery epinephrine C) Epinephrine which has been specifically prepared for administration by injection D) A stabalized form of epinephrine that performs as a sympathetic agonist without the palpatations tremulousness and anxiety side effectsWhich of the following medications is a beta blocker? A) Hydrochlorothiazide B) Lasix C) Atenolol D) Simvastatin Which of the following is a contraindication to the antiarrhythmic Amiodarone? A) Tachycardia B) Allergy to Lidocaine C) Bradycardia D) Hypertension Which of the following is a common side effect of cholesterol medication? A) Muscle pain B) Diabetes C) Headache D) Decreased urination Which of the following most accurately describes the mechanism of action of a "statin"? A) Inhibits platelet aggregation B) Inhibits sodium excretion in the kidneys C) Inhibits cholesterol production in the liver D) Inhibits sodium reabsorption in the loop of Henle Which of the following is an indication to the antiarrhythmic Amiodarone? A) Bradycardia B) Hypercalcemia C) Hypertension D) Supraventricular Tachycardia (SVT)Which of the following is a contraindication to anticoagulant use? A) Recent injury B) Diabetes C) Asthma D) Dizziness Which of the following is an antiplatelet medication? A) Diltiazem B) Labetalol C) Procainamide D) Aspirin A commonly shared action of all antiarrhythmics is that they _____________________. A) Inhibit calcium absorption B) Cause vasoconstriction C) Slow the heart rate D) Prevent bradycardia Which of the following medications is an ACE inhibitor? A) Aspirin B) Labetalol C) Lisinopril D) Simvastatin A Class II antiarrhythmic blocks ____________ channels. A) Potassium B) Beta C) Sodium D) Calcium A potential complication with the administration of a diuretic is ______________________. A) Hypothermia B) Hypernatremia C) Hypokalemia D) Hypoglycemia
- Explain the of side-effects of Tricyclic antidepressants. give an exmaple of the type of Tricylic antidepresents. give a diagram fo the mechansim.Morphine sulfate via intravenous patient-controlled analgesia (PCA) pump is prescribed for a client with severe chronic back pain. Thirty minutes after initiating the PCA pump, the practical nurse (PN) notes that the client has become lethargic. Which action should the PN take? A Reinforce PCA pump teaching. B Check the IV insertion site for swelling. C Determine the client's pain scale rating. D Measure the client's vital signs.What signifies the pharmacodynamic phase when taking acetaminophen (Tylenol) for a headache? Group of answer choices a)breakdown of the tablet form b)relief of headache c)binding of acetaminophen to plasma proteins d)passing of the drug through the kidneys
- Which serotonin agents is NOT acting via interaction with membrane serotonin receptors? a) Ondansetron b) Trazodone c) Sumatriptan d) Citalopram e) Ketanserin Please answer asapA patient will be taking selegiline (Eldepryl), 10 mg daily, in addition to dopamine replacement therapy for Parkinson’s disease. The nurse will implement which precautions regarding selegiline? a )Teach the patient to avoid foods containing tyramine.b )Monitor for dizziness.c )Inform the patient that this drug may cause urine discoloration.d )Monitor for tachycardia and palpitations.



