estion 22 Complete the following statement by choosing one of the pull down answers for the first blank that begin with "1.", and one of the pull down answers for the second blank that begin with "2.": The initial concentration is v the half-life in a second order reaction. This means that as the initial concentration decreases, the half-life 22 of 29
estion 22 Complete the following statement by choosing one of the pull down answers for the first blank that begin with "1.", and one of the pull down answers for the second blank that begin with "2.": The initial concentration is v the half-life in a second order reaction. This means that as the initial concentration decreases, the half-life 22 of 29
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter13: Chemical Kinetics
Section: Chapter Questions
Problem 13.98QE
Related questions
Question
Answer choices
1 inversely proportional to
1 directly proportional to
1 independent of
2 remains constant
2 is longer
2 is shorter

Transcribed Image Text:estion 22
Complete the following statement by choosing one of the pull down answers for the first blank that begin with "1.", and one of the pull down answers for the
second blank that begin with "2.":
The initial concentration is
v the half-life in a second order reaction. This means that as the initial concentration decreases,
the half-life
Question 22 of 29
A Moving to another question will save this response.
..
esc
%23
24
&
3
4
8.
9
delete
E
T
Y
U
enter
A
S
F
G
K
L
ck
return
V
B
alf
alt
trol option
command
option
Expert Solution
Step 1
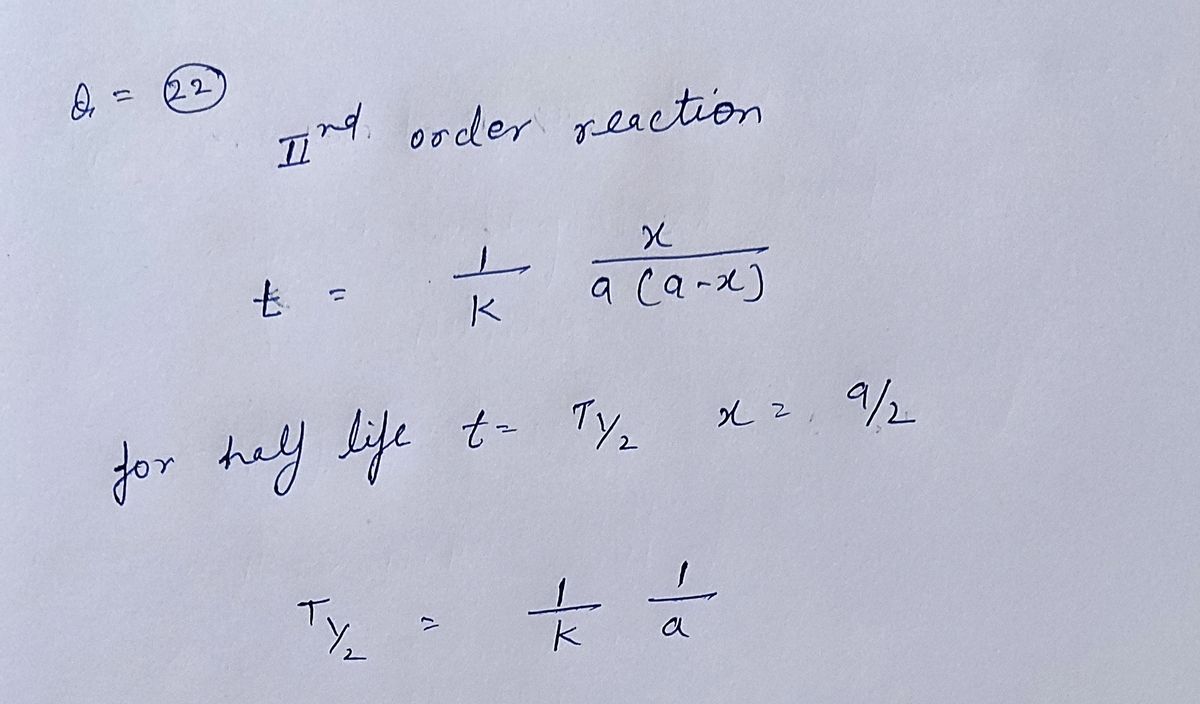
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
