
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
I have 10 min to answer help pls
Transcribed Image Text:CHEM101
101 Chem101
app.101edu.co
Quest
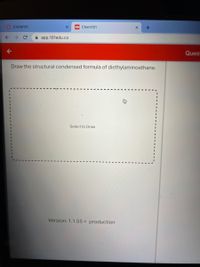
Draw the structural condensed formula of diethylaminoethane.
Select to Draw
Version: 1.1.55+ production
Transcribed Image Text:hem101
+
Question 18 of 20
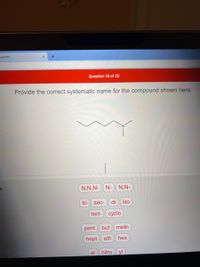
Provide the correct systematic name for the compound shown here.
N,N,N- N- N,N-
tri
sec-
di
iso
tert- cyclo
pent
but
meth
hept eth
hex
al
nitro
yl
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- bard 1 ! New folder M Reading Mode: 1.5... M Gmail ض A- ش X Course Home Z~ is 2 https://openvellum.ecollege.com/course.html?courseld=17485264&OpenVellumHMAC-453c4e9377366ab5782f501c5246fbe0#10001 ▾ 2 Part A alt W= VA 3 Part B Complete previous part(s) S- = Part C Complete previous part(s) Part D Complete previous part(s) X a 2,4-floro, 5-methyl he XI Determine if each of the following cycloalkanes and alkenes can exist as cis-trans stereoisomers. Drag the appropriate items to their respective bins. Cis-trans isomers are possible. Submit Request Answer XO, F3 # 3 E- [» "! D[ LS ra. YouTube 4 C { $ R 24 F1 H₂C. Maps FS ▬▬▬ ▬▬ % 2,4-fluoro,5-methyl h X CH₂ 5 V} TY CH₂ ف GY Br. CH₂ 6 Ą Cis-frans isomers are not possible. XXX YI Copyright © 2022 Pearson Education Inc. All rights reserved. Terms of Use | Privacy Policy Permissions | Contact Us | BY Y 5-Methyl-2,4-hexane X m.__ CH₂CH₂CH-CHF & 7 Hi FB U E W J NI Pearson ی 8 F9 DELL Reset Help Q trimethyl - prt sc 1 ÷ D MA ( 9 к. 30 F10 ن O…arrow_forwardHow do I draw the arrows for each step?arrow_forwardMatch pls, having tough time worn practice problemsarrow_forward
- Ba(NO,l(aq) + K,SOlaq) Baso(s) + 2 KNOlaq) Volume Volume Trial 0.10 M Ba(NO)2 (ml) 0.10 M K,SO, (ml) Mass Baso4 (s) Limiting Reagent 50. 20. 0.46 50. 40. 0.92 3 50. 50. 1.15 4 50. 60. 1.15 50. 80. 1.15 • Question 8 Use the data to identify the limiting reagent for each reaction performed. V Trail 1 a. Neither/Both EV Trial 2 b. Baso4 EV Trial 3 C. KSO4 EV Trial 4 d. Ba(NO3)2 EV Trial 5 e. KNO3 Question Help: Message instructor I Calculator Submit Question • Question 9 Why is the amount of precipitate in the Data Table above the same in Trials 3 through 5? Edit Insert Formats B IVX x' Aarrow_forwardf. e. ? H ㅅㅅ Cu Li (H* workup) 1 EQ Br2 CH3CO2Harrow_forward4-16. Zinc is an essential micronutrient in pet food, but is toxic if present in excess. Do the Zn concentrations (mg/g) for the five cat foods and two dog foods determined by two methods differ significantly at the 95% confidence level? Cat 1 Cat 2 Cat 3 Cat 4 Cat 5 Dog 1 Dog 2 Old method: bris 8 Я 84.9 73.5 173.0 62.7 154.0 80.1 185.0 New faster method: 86.2 81.8 186.0 73.4 138.0 72.5 203.0 SO Souza, S. S. L. Costa, R. G. O. Araujo, C. A. Cats andarrow_forward
- yout References Mailings Review View rt Endnote t Footnote ✓ w Notes es 5 Search Researcher Absorbance 5 4.5 4 3.5 2.5 2 1.5 1 0.5 0 Research 0 Insert Citation 1 1. Construct a standard curve in Excel by plotting concentration (in ppm) on your x-ax (unitless) on your y-axis for your known solutions. Label the axes on the graph and provi title. See Figure 5 in the lab handout for an example. Use a linear trendline to generate a data. Label the graph with the equation and the R2 value. Insert your labeled graph in the Help Manage Sources Style: [APA 2 Bibliography Citations & Bibliography Sciwheel Nitrate Standard Curve 3 4 ty=0.8842x-0.8191 R²=0.9509 Q..**. Concentration Insert Caption 5 6 Insert Table of Figure Update Table Cross-reference Captions ww 7 2. Using the equation from your graph, determine the nitrate concentration (in ppm) in each of samples. Text Predictions: On Accessibility: Investigate O Search 8509 FORGE *arrow_forwardDrowser Time Limit: 0:20:00 Time Left:0:15:19 Glenton Morris: Attempt 1 160 40 3.0 Page 1: 140 - 35 3.0 120 10 25 100 2.0 80 60 Z 10 Page 2: 05- 20 0 2. Time is 10 Timetsk 10 Page 3: Of all the graphs shown above, what plot type is correctly represented? O second order Page 4: O no plot shown is valid 4. 1st order 3rd order OL X (W) FONarrow_forwardQuestion 1 of 20 A barrel of crude oil has a volume of 42 gallons, only approximately 45% of which is processed into gasoline. If your car achieves 32 mi/gal, and you drive 36,000 miles in one year, how many barrels of crude oil are required to run your car for a year? Tap here or pull up for additional resourcesarrow_forward
- Search he web.. H Take T X G What is th P Pearson S b Answered M Inbox (1,0 er 1 0 8 https://bblearn.rcsj.edu/webapps/assessment/ Question Completion Status: 3. 50 70 100 110 120 130 14G 18 QUESTION 3 3.478 g x 1.164 g What is the best answer to report for -0.116 g/mL g/mL? 2.00 mL O a. 1 g/mL O b.1.904 g/mL Oc.1.90478009 g/mL O d. 1.9 g/mL Oe1.91 g/mL QUESTION 4 The state of matter for an object that has both definite volume and definite shape i a. gaseous state b. elemental state OC. solid state Od. mixed state e. liquid state Click Save and Submit to scue and subrt CackSae74nswers to save al cnswers. Type here to searcharrow_forwardneed help with both questions pls & thank you!!arrow_forwardNeed help with homeworkarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY