
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Create a graph in excel displaying the relationships identified in the data tables. The graph is numbered properly, has appropriate titles and legends, and the axes of the graph are properly labeled with units. The appropriate tables are referenced so that the reader can see which graph is based on which data table. Make sure to include R2 values, and th equation for the line.
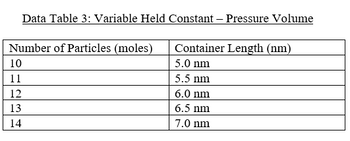
Transcribed Image Text:Data Table 3: Variable Held Constant - Pressure Volume
Number of Particles (moles)
10
11
12
13
14
Container Length (nm)
5.0 nm
5.5 nm
6.0 nm
6.5 nm
7.0 nm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Just #2 pleasearrow_forwardA student prepares a 0.38 mM aqueous solution of acetic acid (CH3CO₂H). Calculate the fraction of acetic acid that is in the dissociated form in his solution. Express your answer as a percentage. You will probably find some useful data in the ALEKS Data resource. Round your answer to 2 significant digits. 11 % 0.0 x10 X S 0 Earrow_forwardA certain liquid X has a normal boiling point of 105.70°C and a boiling point elevation constant =Kb2.16·°C·kgmol−1. A solution is prepared by dissolving some potassium bromide (KBr) in 400.g of X. This solution boils at 107.3°C. Calculate the mass of KBr that was dissolved. Round your answer to 2 significant digits. ____garrow_forward
- Measurement and Matter Adding or subtracting and multiplying or dividing measurements 1/3 B A chemistry student must write down in her lab notebook the concentration of a solution of sodium hydroxide. The concentration of a solution equals the mass of what's dissolved divided by the total volume of the solution. Here's how the student prepared the solution: The label on the graduated cylinder says: empty weight: 7.2 g . She put some solid sodium hydroxide into the graduated cylinder and weighed it. With the sodium hydroxide added, the cylinder weighed 63.517 g. . She added water to the graduated cylinder and dissolved the sodium hydroxide completely. Then she read the total volume of the solution from the markings on the graduated cylinder. The total volume of the solution was 91.0 mL. What concentration should the student write down in her lab notebook? Be sure your answer has the correct number of significant digits. g-mL Garrow_forwardlculation questions require you to enter a numerical answer. You should not include units in your reported an ough you should note them in your calculations. Unless the problem specifically ask for an answer in the pro significant figures, it is often in your best interest to enter as many digits as your calculator will give you. 1 Question 1 Given the unbalanced chemical equation, C5H12(1) + O2(g) → CO(g) + H20(1), which one of the following correctly expresses the mole ratio between CO and oxygen? 5 mole CO/11 mole O2 O 1 mole CO/1 mole O2 O 10 mole CO/11 mole O2 O 10 mole CO/6.5 mole O2 Question 2arrow_forward4b. Convert the mass of the graduated cylinder with water using an analytical balance from grams to tons. 98.5107 g 4 4 13.8 mL 13.8 mL X 4 X 4 5. Beginning with the volume of water measured by the graduated cylinder, convert the volume from milliliters to liters and microliters. 4 4 4 P 4 15 15 L (rounded) HL 14 tonsarrow_forward
- For the reaction 2A(g) s B(g) K. = 69 For the reaction A(g) = ½ B(g) Kc = For the reaction ½ B(g) A(g) Kc = Report each answer to two significant figures. Do NOT use scientific notation.arrow_forwardA chemist adds 305.0 mL of a 0.41 M barium chloride (BaCl₂) solution to a reaction flask. Calculate the millimoles of barium chloride the chemist has added to the flask. Round your answer to 2 significant digits. 1.25 x 10 mmol [NN] X 5 ? Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility Ad DII F11 F12 F10 FO FB F7 esc ! 1 Explanation 2 Check 39: F2 14/ # 3 80 F3 $ 4 000 F4 R % 5 ܐ FS T ^ 6 F6 Y & 7 U * 00 8 ہ ۔ 9 ) 0 0 P + 11 T D olo Ararrow_forwardWhy is it inappropriate to report yields with decimal-place accuracy (as opposed to whole numbers)?arrow_forward
- One half tsp of table salt (NaCl) weighs aproximately 2.0 grams. (You will need to look up the molar mass for NaCl) There are approximately 237 ml in a cup. Determine the Molarity of the solution 1 cup water with one half tsp table salt. Determine the molarity of this solution using the formula M=g/MM/L. What is the molarity? Show your calculations. (Remember to convert 237 mL to L)arrow_forwardA solution of phosphoric acid is sometimes used to acidify foods, such as jams, to give them a tangy or sour taste. Calculate the amount of phosphoric acid, in moles, that is in 32.8 mL of a 4.46 mol/L phosphoric acid solution. Record your numerical answer with the correct number of significant digits in the first answer box. You do not need to include units as the units appear for you beside the answer box already.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY