Consider the mechanism of enolase, as indicated below. Which of the following correctly describes the roles of the Mg2+ as illustrated in the figure? (This is a multi- select question). Mg2+ Mg2 Enolase po²- OH -C-C-H H OH НО. H-N-H Lys 345 0 Glu211 2-Phosphoglycerate bound to enzyme Mg2+ Mg2+ PO²- OH -C-H OH HO H H-N+-H Lys 345 Glu211 Enolic intermediate HOH PO3- Phosphoenolpyruvate The metal ion (Mg2+) is helping to stabilize the extra negative charge that developed on the carboxyl group in the enolic intermediate. The metal ion (Mg2+) is serving as a general base, removing a proton in order to improve the quality of the nucleophile. | The metal ion (Mg²+) is assisting in the oxidation of the carboxyl carbon through metal ion catalysis. The metal ion (Mg2+) is helping to orient the substrate properly in the active site. The metal ion (Mg2+) is accepting a proton in order to improve the quality of the leaving group.
Consider the mechanism of enolase, as indicated below. Which of the following correctly describes the roles of the Mg2+ as illustrated in the figure? (This is a multi- select question). Mg2+ Mg2 Enolase po²- OH -C-C-H H OH НО. H-N-H Lys 345 0 Glu211 2-Phosphoglycerate bound to enzyme Mg2+ Mg2+ PO²- OH -C-H OH HO H H-N+-H Lys 345 Glu211 Enolic intermediate HOH PO3- Phosphoenolpyruvate The metal ion (Mg2+) is helping to stabilize the extra negative charge that developed on the carboxyl group in the enolic intermediate. The metal ion (Mg2+) is serving as a general base, removing a proton in order to improve the quality of the nucleophile. | The metal ion (Mg²+) is assisting in the oxidation of the carboxyl carbon through metal ion catalysis. The metal ion (Mg2+) is helping to orient the substrate properly in the active site. The metal ion (Mg2+) is accepting a proton in order to improve the quality of the leaving group.
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 18P: Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in...
Related questions
Question
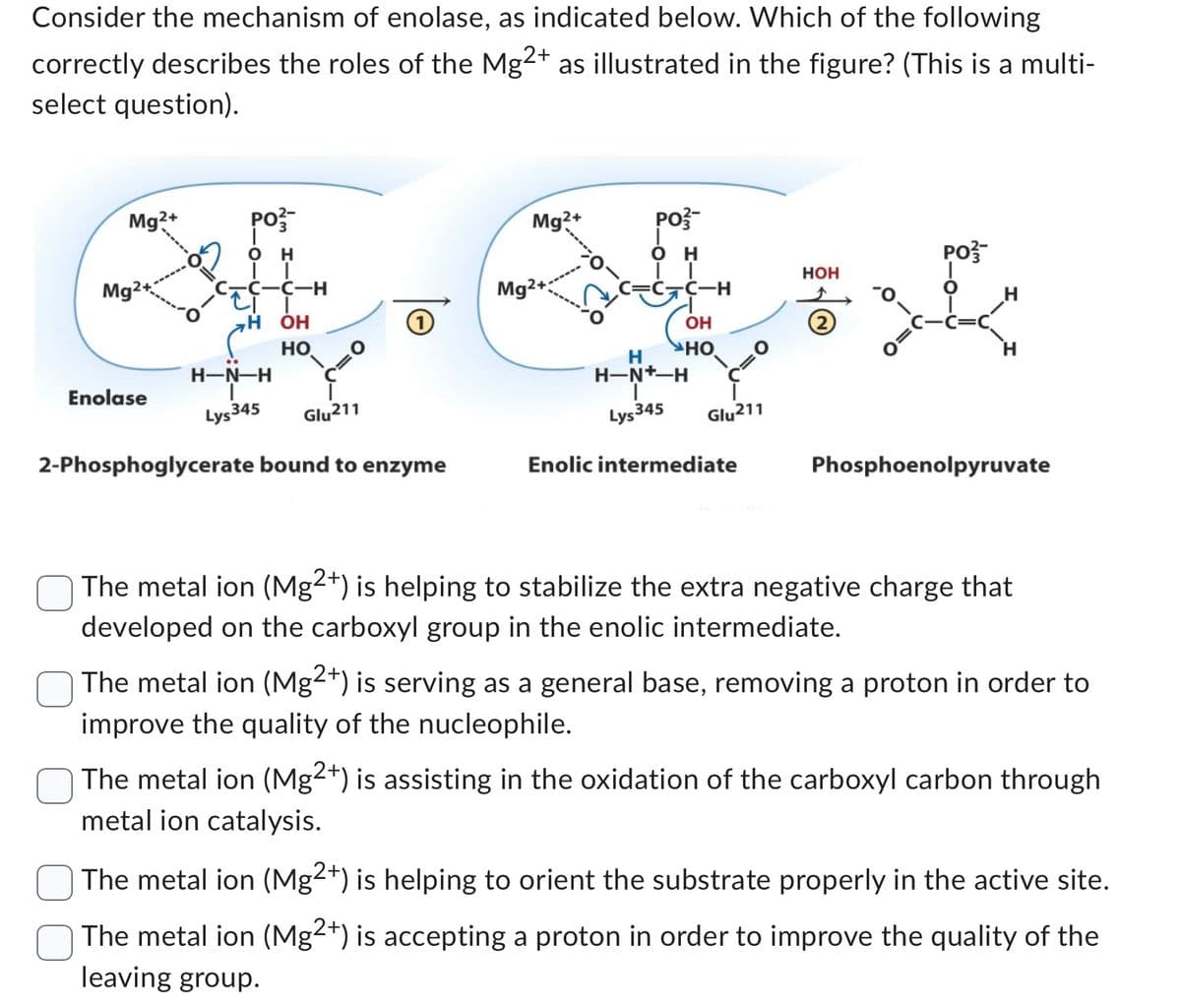
Transcribed Image Text:Consider the mechanism of enolase, as indicated below. Which of the following
correctly describes the roles of the Mg2+ as illustrated in the figure? (This is a multi-
select question).
Mg2+
Mg2
Enolase
PO3-
OH
-C-C-H
H OH
HO
H-N-H
Lys 345
Glu211
2-Phosphoglycerate bound to enzyme
Mg2+
Mg2
PO3-
OH
C-C-H
OH
HO
H
H-N*-H
Lys 345
O
Glu211
Enolic intermediate
HOH
PO3-
H
Phosphoenolpyruvate
The metal ion (Mg2+) is helping to stabilize the extra negative charge that
developed on the carboxyl group in the enolic intermediate.
The metal ion (Mg2+) is serving as a general base, removing a proton in order to
improve the quality of the nucleophile.
The metal ion (Mg2+) is assisting in the oxidation of the carboxyl carbon through
metal ion catalysis.
The metal ion (Mg2+) is helping to orient the substrate properly in the active site.
The metal ion (Mg2+) is accepting a proton in order to improve the quality of the
leaving group.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning