
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
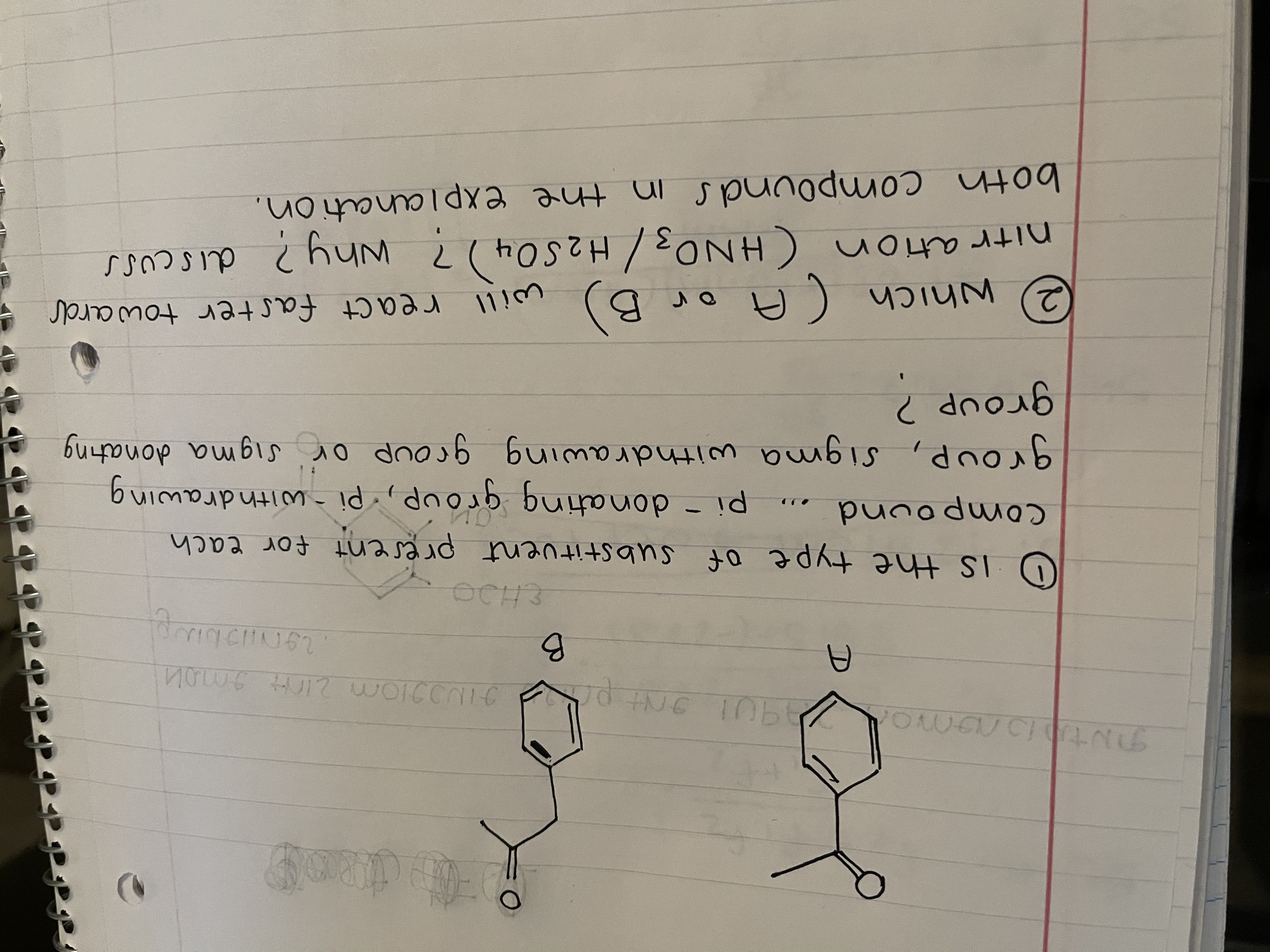
Look at image please
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculate delta H for the graph below. y = -59.88x -1.6547 Pen -1.84 188003100032 -1.85 -1.805 -1.86 -1.005 -1.87 1875 Ink vs 1/T 0.0033 0.0034 0.0035 0.0036 00037 ya-59 885x-1,6547 R² -0.9000 1/T (10) N What is the concentration of Magnesium in 20mL of water if 35mL of 0.1M EDTA is used to titrate?arrow_forwardFile Edit View History Bookmarks Profiles Tab Window Help O ? Q : hrome N Watch Gilmore Girls | Netflix a ALEKS A ALEKS - Reyna Garcia - Learn OPsychology Research Sign-Up x T WPAL 101 233_Spring 2022 A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IvdWKW_BBZZI6tTytly4Fcfu6zOtOf8oMM9sfQgW6H_IIXnxiNhBSpKS52uqVQ2d_u6pd3.. Spotify Web Playe. M Common Ethical D. O CHEMICAL REACTIONS 1/5 Solving for a reactant using a chemical equation Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,) from the air to form water (H,0) and acetic acid (CH,COOH), the main ingredient of vinegar. What mass of acetic acid is produced by the reaction of 1.9 g of oxygen gas? Be sure your answer has the correct number of significant digits. IIarrow_forwardne File Edit View History Bookmarks Profiles Tab Window Help Watch Gilmore Girls | Netflix a Dashboard A ALEKS - Reyna Garcia O Psychology Research Sign-Up x T WPAL 101 233 Spring 2 A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH-IvdWKW_BBZZ16tTytly4Fcfu6zOtOf80MM9smRgOXk4zql8rHN-B4Sv7Er8YkAkkZH6IMU.. O ☆ Spotify Web Playe.. M Common Ethical D.. O CHEMICAL REACTIONS Solving moles-to-moles limiting reactant problems 1/5 Solid aluminum (Al)and oxygen (02) gas react to form solid aluminum oxide (Al,03). Suppose you have 2.0 mol of Al and 7.0 mol of O, in a reactor. Suppose as much as possible of the Al reacts. How much will be left? Round your answer to the nearest 0.1 mol. I molarrow_forward
- Chrome File Edit View History Bookmarks Profiles Tab Window Help 91% O Sun 10:58 O St. John's University - My Appl x A ALEKS - Iffat Khan - Knowledg x + i www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLIq7wcPWKZBYGfE9IMFjeRiThHre2U30XGpg1_cQJKNFZspY-BJWbSypPLYdk. Knowledge Check Question 8 Iffa The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute temperature and the gas constant R. Suppose the osmotic pressure of a certain solution is measured to be 11. atm at an absolute temperature of 349. K. Write an equation that will let you calculate the molarity c of this solution. Your equation should contain only symbols. Be sure you define each symbol other than R. Your equation: Definitions of your symbols: D= 11. atm = 349. K IIarrow_forwardChrome File Edit View History Bookmarks Profiles Tab Window Help N Watch Gilmore * Dementia Frien x 13 unread) - dte x T Lobby | Top Hat x 2 ALEKS A ALEKS - Reyna > A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZ16tTytly4Fcfu6zOtOf80MM9s7G0XWajIKntF38TIGBGK Apps Spotify Web Playe. M Common Ethical D. O THERMOCHEMISTRY Understanding the definitions of heat and work A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gaseous products. The cylinder has a piston that moves in or out, as necessary, to keep a constant pressure on the mixture of 1 atm. The cylinder is also submerged in a large insulated water bath. (See sketch at right.) From previous experiments, this chemical reaction is known to release 133. kJ of energy. The position of the piston is monitored, and it is determined from this data that the piston does 116. kJ of work on the system during the reaction. O exothermic Is the reaction exothermic or endothermic?…arrow_forwardChrome File Edit View History Bookmarks Profiles Tab Window Help N Watch Gilmore Girls | Netflix DashooardS A ALEKS - Reyna Garcia - Learn x A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZI6tTytly4Fcfu6zOtOf80MM9s8-pEjBx4anm2FVArDlj7Hnm8sjOBpH_25U... Apps Spotify Web Playe. M Common Ethical D. O CHEMICAL REACTIONS Writing a chemical equation from a description of the reaction 0/5 Solid iron(II) sulfide reacts with aqueous hydrochloric acid (HCI) to produce aqueous iron(II) chloride and hydrogen sulfide gas (H,S). Write a balanced chemical equation for this reaction. O-0 Explanation Check O 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use 2 MacBook Airarrow_forward
- Ask Laftan Anlamaz - Episode St. John's University - My App X A ALEKS - Iffat Khan - Learn G In valence electrc -> A www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-lijkPWvZoZLgkt1FLIq7wcPWKzBYGfE9IMFj8s1-UE-m2k5S O MATTER Finding the side length of a cube from its volume in liters A technical machinist is asked to build a cubical steel tank that will hold 390 L of water. Calculate in meters the smallest possible inside length of the tank. Round your answer to the nearest 0.01 m. Omarrow_forwardrome File Edit View History Bookmarks Profiles Tab Window Help N Watch Gilmore x (5 unread) - dtE X AALEKS A ALEKS - David A ALEKS - Reyna A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZI6tTytly4Fcfu6zOtOf8oMM9sv O Spotify Web Playe.. M Common Ethical D.. O CHEMICAL REACTIONS Interconverting number of atoms and mass of compound Calculate the number of gold atoms in a 150.0 g sample of gold(III) chloride (Au,Cl). Be sure your answer has a unit symbol if necessary, and round it to 4 significant digits. Explanation Check APR 18 IIarrow_forwardWhich layer was the ether layer in the simulator? NB: ether is shorthand for diethyl ether. Top layer Bottom layerarrow_forward
- Chrome File Edit View History Bookmarks Profiles Tab Window Help Watch Gilmore Girls x * Dementia Friend Ce X y! Yahoo Lobby Top Hat www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IvdWKW_BBZZI6tTytly4Fcfu6zOtOf8oMM9s Apps Spotify Web Playe... M Common Ethical D... O CHEMICAL REACTIONS Interconverting number of atoms and mass of compound Calculate the number of magnesium atoms in a 80.0 g sample of forsterite (Mg, SiO4). Be sure your answer has a unit symbol if necessary, and round it to 3 significant digits. Explanation Check IIarrow_forwardme File Edit View History Bookmarks Profiles Tab Window Help N Watch Gilmore Girls | Netflix a ALEKS A ALEKS - Reyna Garcia - Learn X O Psychology Research Sign-Up x T WPAL 101233 Spring 2022 www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IvdWKW_BBZZ16tTytly4Fcfu6zOtOf8oMM9sqG0YAPcLeyNqu8U3rAMkvKt93Vpt7xcKoyt... O * O Spotify Web Playe.. M common Ethical D. O CHEMICAL REACTIONS R 0/5 Solving for a reactant using a chemical equation A major component of gasoline is octane (C,H18). When octane is burned in air, it chemically reacts with oxygen gas (O,) to produce carbon dioxide (CO,) (H,). and water What mass of oxygen gas is consumed by the reaction of 4.84 g of octane? Be sure your answer has the correct number of significant digits. Ac Check Explanation O 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center IIarrow_forwardEdit View History Bookmarks Profiles Tab Window Help ome File d Dashboard ALEKS - Reyna Garcia - Lear Watch Gilmore Girls | Netflix A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZ16tTytly4Fcfu6zOtC Spotify Web Playe... M Common Ethical D... O CHEMICAL REACTIONS Writing a chemical equation from a description of the reaction Solid potassium and chlorine gas combine to produce solid potassium chloride. Write a balanced chemical equation for this reaction. Explanation Check FEB 2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY