
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
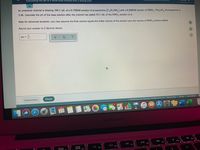
Transcribed Image Text:Calculating the pH of a weak base titrated with a strong acid
An analytical chemist is titrating 104.1 mL of a 0.1200M solution of propylamine (C,H,NH,) with a 0.2600M solution of HNO3. The p K, of propylamine is
3.46. Calculate the pH of the base solution after the chemist has added 53.1 mL of the HNO, solution to it.
Note for advanced students: you may assume the final volume eguals the initial volume of the solution plus the volume of HNO, solution added.
Round your answer to 2 decimal places.
pH =
Explanation
Check
O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use
Privacy
| Accessibility
APR
23
MacBook Air
O00
000 F4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A chemistry graduate student is given 100. mL of a 0.40M nitrous acid (HNO,) solution. Nitrous acid is a weak acid with K 4.5 x 10. What mass of -4 NaNO, should the student dissolve in the HNO, solution to turn it into a buffer with pH =3.16? %3D You may assume that the volume of the solution doesn't change when the NaNO, is dissolved in it. Be sure your answer has a unit symbol, and round it to 2 significant digits. x10 Submit Assignment Continue 2021 McGraw Hill LLC. AlI Rights Reserved. Terms of Use Privacy Center | Accessibility MacBook Air F11 Figarrow_forwardAn analytical chemist is titrating 152.0 mL of a 0.2100 M solution of benzoic acid (HCH,CO,) with a 0.6700 M solution of NaOH. The p K, of benzoic acid is 4.20. Calculate the pH of the acid solution after the chemist has added 29.70 mL of the NaOH solution to it.arrow_forwardAspirin (C 9H 8O 4) is a weak monoprotic acid (K a = 3.3 x 10-4). You are analyzing a samplefor a pharmaceutical company with a quantitative titration and dissolve 10 tablets, eachcontaining 200 mg of aspirin, in 150 mL of water.a) Determine the pH of this solution.b) Determine the pH of the solution after you have added 20.0 mL of 0.500 M NaOH.arrow_forward
- A 1.0-L buffer solution contains 0.100 mol acid, HA, and 0.100 mol base, NaA. The value of K, for the acid HA is 1.8x104. Ignoring the small volume change, what is the most likely pH of the solution if a 10.0 mL- sample of 1.00M HCI is added to the original buffer? O 1.20 < pH < 1.45 O 5.50 < pH < 5.74 O 4.10 < pH < 4.22 O 3.51 < pH < 3.69arrow_forwardFor a buffer initially containing .08 mol of benzoic acid (HC7H5O2) and .12 mol of C7H5O2 in 1.0 L, what is the pH after .07 mol of OH is added to the buffer? .07 mol OH- .08 mol HC,H,O2 .12 mol C,H¿0; O 4.57 O 5.47 O 2.91 O 4.37 O 3.71arrow_forward4. What is the pH at each of the following points in the titration of 25.00 mL of 0.100 M CH,CH;COOH with 0.100 M NaOH? a) before the addition of any NaOH; b) after the addition of 10.00 mL of 0.100 M NaOH; c) after the addition of 12.50 mL of 0.100 M NaOH; d) after the addition of 25.00 mL of 0.100 M NaOH; e) after the addition of 26.00 mL of 0.100 M NaOH.arrow_forward
- 2. Suppose 1.44 0.150 M benzoic acid solution. Calculate the pH of the resulting buffer solution as- suming the volume of the solution does not change on addition of the solid. of sodium benzoate, Na+C,H3COO-, is added to 100.0 mL of thearrow_forward1000 mL of a buffer solution contains 0.275 moles of HCN, and 0.175 moles of NaCN. Ka of HCN = 6.2 x 10^-10.a) calculate the initial pH b) calculate the pH of the buffer after the addition of 27.5 L of 1.0 M of HCl. c) what is the change in pH of the solution if the same amount of Hcl which was used in part (b) were added to 1000 mL pure water.arrow_forwardA buffer is made using 100.0 mL of 0.100 M CH3 CH2 COOH (propanoic acid) and 100.0 mL of 0.100 M NACH3 CH2 COO (sodium propanoate). A) Explain in your own words what will occur (at the molecular level) when an nitric acid is added to the buffer? What would be the effect on the pH? B) Explain in your own words what will occur when LIOH is added tot he buffer? What would be the effect on the [H+]?arrow_forward
- Payliarrow_forwarddont hand writingarrow_forwardAn analytical chemist is titrating 185.8 mL of a 1.200M solution of benzoic acid (HC,H,CO,) with a 1.200M solution of KOH. The p K, of benzoic acid is 4.20. Calculate the pH of the acid solution after the chemist has added 196.6 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added. Round your answer to 2 decimal places. olo pH = ] Ar G Explanation Check © 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility ............................... .................................. .............. .... .arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY