
College Physics
11th Edition
ISBN: 9781305952300
Author: Raymond A. Serway, Chris Vuille
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
A gas is taken through the
J
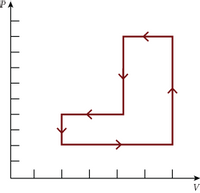
Transcribed Image Text:V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Calculate the efficiency of a heat engine operating between two reservoirs at temperatures T₁ = 400 K and T₂ = 300 K. Select one: O a. η = 0.25 - Ο b. η = 3.0 O c. n = 4.0 O d. n = 0.33333333333333arrow_forwardA gas expands from 2.2 L to 3.6 L against a constant external pressure of 1.6 atm. What is the work done? A. -227 J B. 227 J C. 2.24 J D. -2.24 Jarrow_forwardThere are 2 kg of gas with R = 400 J/kg- oK and k = 1.25 that undergo a nonflow constant volume process from p1 = 600 kPa and T1 = 87 oC to p2 = 1600 kPa. During the process, the gas is internally stirred and there are added 220 kJ of heat. Find, a.T2 in oC b. Work input c. change of Entropy, Sarrow_forward
- A heat engine does 30.0 J of work and exhausts 35.0 J of waste heat during each cycle. Part A What is the engine's thermal efficiency? Express your answer as a percentage. 46.2 % Submit Correct Part B Previous Answers If the cold-reservoir temperature is 30.0 °C, what is the minimum possible temperature in °C of the hot reservoir? Express your answer in degrees Celsius. —| ΑΣΦ 383.35 ? °Carrow_forwardI Rev Part A How much work is done by the gas in the process shown in the figure? (Figure 1) Express your answer in joules. V AZO ? W = J Submit Request Answer Figure 1 of 1 > Provide Feedback Р (КРа) 400- 200 i V (cm³) 100 200 300arrow_forwardFind the amount of work W done on the gas during the entire process? express answer in terms of some or all of the variables p and V W = ?arrow_forward
- In an engine, an almost ideal gas is compressed adiabatically to half its volume. In doing so, 2710 J of work is done on the gas. Part A ▼ How much heat flows into or out of the gas? Express your answer with the appropriate units. ? Q = Value Units Submit Request Answer Part B What is the change in internal energy of the gas? Express your answer with the appropriate units. HẢ ? AU = Value Units Submit Request Answerarrow_forwardAn ideal gas, at initial temperature T1 and initial volume 3.1 m3, is expanded adiabatically to a volume of 6.8 m³, then expanded isothermally to a volume of 17 m3, and then compressed adiabatically back to T1. What is its final volume? Number i Unitsarrow_forwardanswer the question >>>>>arrow_forward
- Can you help with "c" was told to use as less digits as possible 3. An unknown number of moles of an ideal monoatomic gas expand reversibly from Vi = 3.10 m3 to Vf = 4.00 m3, at a constant pressure of 1.47 atm and an initial temperature of 300 K. a. Find the number of moles of gas. 185b. Find the final temperature of the gas K. 387 c. Calculate the work done by the gas. 134053 J <<<< this is the wrong answer. (should use less digits for this answer)arrow_forwardA thermodynamic process can be represented using a PV diagram where the pressure and volume at every stage is recorded. In the thermodynamic process shown below the work done by the system is РА A O the area under the PV curve and positive the area under the PV curve and negative B is the product of the change in volume during the process and the change in pressure. cannot be found from the PV grapharrow_forward280.5 K is not correct. Please look againarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON