
Chemistry: Matter and Change
1st Edition
ISBN: 9780078746376
Author: Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher: Glencoe/McGraw-Hill School Pub Co
expand_more
expand_more
format_list_bulleted
Concept explainers
Question

Transcribed Image Text:5.
PbSO4 →
PBSO3 +
02
Туре of reaction:
12ラー
N2l6 +
6.
NH3 +
H2
Type of reaction:
7.
H20 +
SO3 >
H2SO4
Type of reaction:
8.
H2SO4 +
NH40H →
Н20 +
(NH4)2SO4
Type of reaction:
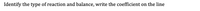
Transcribed Image Text:Identify the type of reaction and balance, write the coefficient on the line
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A student finds that 37.80mL of a 0.4052MNaHCO3 solution is required to titrate a 20.00-mL sample of sulfuric acid solution. What is the molarity of the acid? The reaction equation is H2SO4+2NaHCO3Na2SO4+2H2O+2CO2.arrow_forwardWrite a chemical equation representing the decomposition of water into two gaseous products. What are the products?arrow_forwardThe Solvay process is multistep industrial method for the manufacture of sodium carbonate, Na2CO3, which is also known as washing soda. Although most of the industrialized world utilizes the Solvay process for production of sodium carbonate, it is not manufactured in the United States because it can be obtained at lower cost from a large natural deposit in Wyoming. Questions 19 through 22 are based on reactions in that process. Sodium carbonate is used for many purposes, including glass manufacturing. How much NaCl is needed to react completely with 83.0g of ammonium hydrogen carbonate in NaCl+NH4HCO3NaHCO3+NH4Cl?arrow_forward
- Apply Describe the reaction of aqueous solutions of sodium sulfide and copper(ll) sulfate, producing the precipitate copper(ll) sulfide.arrow_forwardIn a dilute solution of sodium chloride in water, the sodium chloride is the: a. solvent. b. solute. c. precipitate. d. reactant.arrow_forwardCertain vegetables and fruits, such as potatoes and apples, darken quickly when sliced. Submerging the slices in water slows this process. Explain.arrow_forward
- Apply Students conducted a lab to investigate limiting and excess reactants. The students added different volumes of sodium phosphate solution (Na3PO4) to a beaker. They then added a constant volume of cobalt(ll) nitrate solution (CO(NO3)2) stirred the contents, and allowed the beakers to sit overnight. The next day, each beaker had a purple precipitate at the bottom. The students decanted the supernatant from each beaker, divided it into two samples, and added one drop Of sodium phosphate solution to one sample and one drop of cobalt(ll) nitrate solution to the second sample. Their results are shown in Table 11.5. a. Write a balanced chemical equation for the reaction. b. Based on the results, identify the limiting reactant and the excess reactant for each trial.arrow_forwardAn analytical procedure for finding the chloride ion concentration in a solution involves the precipitation of silver chloride: Ag++ClAgCl. What is the molarity of the chloride ion if 16.80mL of 0.629M silver nitrate the source of silver ion is needed to precipitate all of the chloride ion in a 25.00-mL sample of the unknown? Silver nitrate solution is added to an unknown chloride-ion solution, yielding a precipitate of silver chloride.arrow_forwardA solution containing 1.63g of barium chloride is added to a solution containing 2.40g of sodium chromate chromate ion, CrO42. Find the mass in grams of barium chromate that can precipitate. Also determine which reactant was in excess, as well as the mass in grams in excess of the amount required by the limiting reactant.arrow_forward
- Complete the missing information in the following skeletion equation and balance the chemical equation: NaOH(aq)+3NaCl(aq)+Al(OH)3(aq)arrow_forwardA student weighs out a 4.80-g sample of aluminum bromide, transfers it to a 100-mL volumetric flask, adds enough water to dissolve it, and then adds water to the 100-mL mark. What is the molarity of aluminum bromide in the resulting solution?arrow_forwardA student was given a 1.6240-g sample of a mixture of sodium nitrate and sodium chloride and was asked to find the percentage of each compound in the mixture. She dissolved the sample and added a solution that contained an excess of silver nitrate. The silver ion precipitated all of the chloride ion in the mixture as silver chloride. It was filtered, dried, and weighed. Its mass was 2.056g. What was the percentage of each compound in the mixture?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning  World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
