
Concept explainers
Interpretation:
Based on the structure given, the different types of stereoisomers for each of the conditions given are to be identified.
Concept introduction:
A chiral carbon atom is attached to four different atoms or groups of atoms. The number of stereoisomers is determined by using the formula
Stereoisomers arise from chiral centers as well as the arrangement of atoms or groups attached to the double bonded carbon atoms.
The pair of isomers designated
Enantiomers are non-superimposable mirror images of each other.
Stereoisomers that are not mirror images are diastereomers.
Answer to Problem 35P
Solution:
a) The number of stereoisomers represented by the given constitution is
b) If the substituents on the five-membered ring are cis to each other, then there are four stereoisomers represented by the given constitution.
c) If the butenyl side chain has the Z configuration of its double bond, the given constitutional isomer has four possible stereoisomers.
d) Stereochemically accurate representations of all the stereoisomers are as follows:
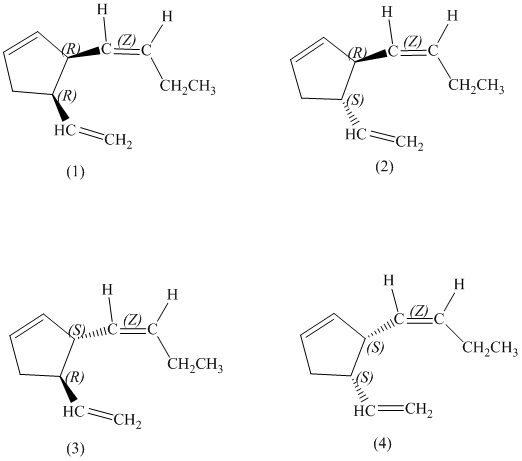
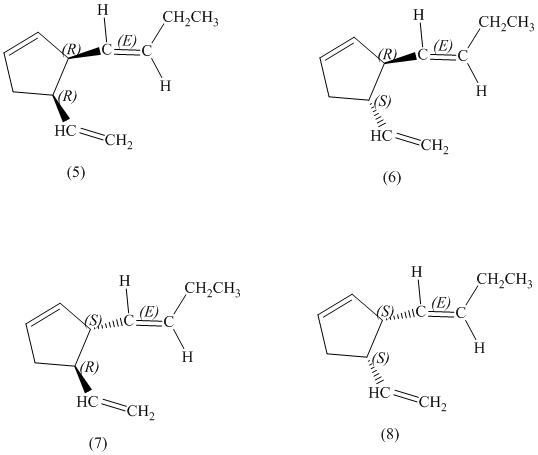
e) The pairs of enantiomers are as follows:
The pairs of diastereomers are as follows:
Explanation of Solution
a)
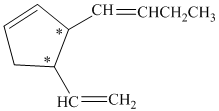
In the given structure, there are two chiral carbon atoms indicated by
Also, the double bond of butenyl side chain can form E and Z isomers, so the total number of possible stereoisomers will be:
Therefore, the number of stereoisomers represented by the given constitution is
b)
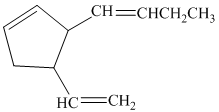
So, if the substituents on the five-membered ring are cis to each other, then the double bond in the butenyl side chain can be in the E or Z form. Thus, there are
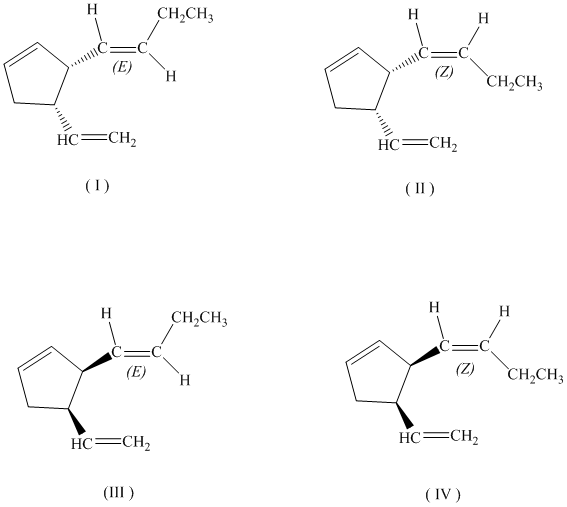
c)

If the butenyl side chain has the Z configuration of its double bond, the two substituents on the ring can be oriented in four different ways. The number of possible stereoisomers is
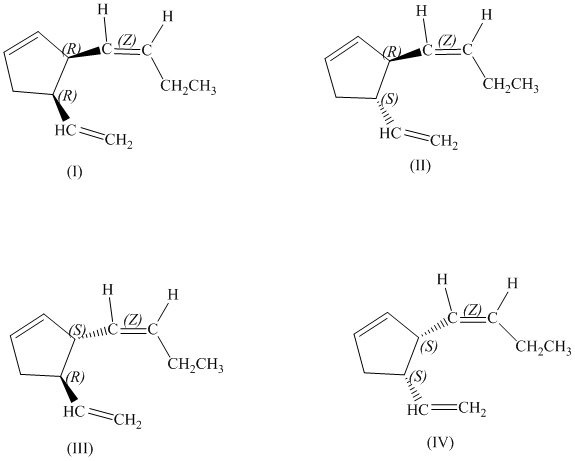
d)

There are total
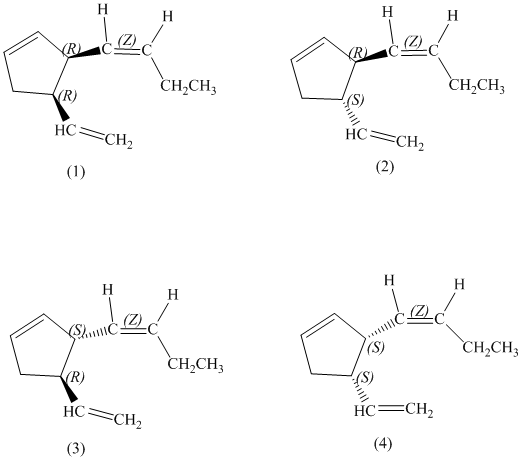
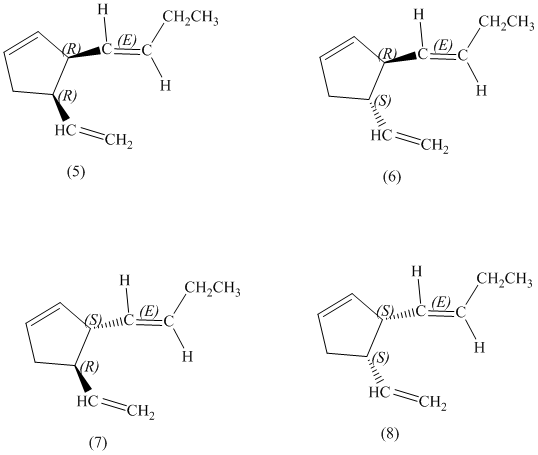
e)
Enantiomers are non-superimposable mirror images of each other. Diastereomers are the stereoisomers which are not mirror images.
The pairs of enantiomers are as follows:
The pairs of diastereomers are as follows:
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry - Standalone book
- Isotretinoin is a retinoid derivative of vitamin A used in the treatment of severe recalcitrant acne. What is the degree of substitution of the encircled alkene functional group in its structure? HO, A trisubstituted monosubstituted tetrasubstituted D disubstitutedarrow_forwardFill in the boxes with the correct organic structure. You must show relative stereochemistry where necessary. You only need to draw one enantiomer if the product is a racemic mixture. 1. OsO4 2. NaHSO3 MCPBA HglO6 1. NaOH 2. HCIarrow_forwardWhen bromine is added to two beakers, one containing phenyl isopropyl ether and the other containing cyclohexene, the bromine color in both beakers disappears. What observation could you make while performing this test that would allow you to distinguish the alkene from the aryl ether?arrow_forward
- Benzopyrene, naphthalene and pyrene are members of these group of aromaticcompounds: *Non-benzenoid aromatic compoundsHeterocyclic aromatic compoundsBenzenoid aromatic compoundsHeteronuclear compoundsWhat type of aromatic compound is pyridine? *Benzenoid aromatic compoundNon-benzenoid aromatic compoundHomonuclear cyclic compoundHeterocyclic aromatic compoundWhat property of aromatic rings prevent the involvement of the conjugated structure toaddition reactions? *Radical stabilizationResonance stabilityInductive effectAromatic effectarrow_forwardCompound X, C8H17Cl, is a chiral product of the radical chlorination of 4-methylheptane.X reacts in SN2 fashion with NaI in acetone to form Z, C8H17I. When the reactant is the R-enantiomer of X, only the R-enantiomer of Z is formed.Draw a structural formula for X; do not show stereochemistry.arrow_forwardWrite structural formulas for all aldehydes with the molecular formula C6H12O and give each its IUPAC name. Which of these aldehydes are chiralarrow_forward
- Only three stereoisomers are possible for 2,3-dibromo-2, 3-dichlorobutane. Draw them, indicating which pair are enantiomers (optical isomers). Why does the other isomer not have an enantiomer?arrow_forwardChlordane, like DDT, is an alkyl halide that was used as an insecticide for crops such as corn and citrus and for lawns. In 1983, it was banned for all uses except against termites, and in 1988, it was banned for use against termites as well. Chlordane can be synthesized from two reactants in one step. One of the reactants is hexachlorocyclopentadiene. What is the other reactant?arrow_forwardHow many different organic products are possible when pentan-2-ol is heated in the presence of acid? Ignore any stereoisomers (cis/trans isomers).arrow_forward
- A chemist isolated an aromatic compound with molecular formula C6H4Br2. He treated this compound with nitric acid and sulfuric acid and isolated threedifferent isomers, in different amounts, with molecular formula C6H3Br2NO2. What was the structure of the original compound?arrow_forwardWhen the acyclic portion of the molecule contains more carbon atoms (or an important functional group), name the ring as a cycloalkyl substituent. (g) xe (1) (h)arrow_forwardDraw the two major organic products of the reaction in ether. Show the stereochemistry of the products. Are the products single enantiomers or racemic mixtures?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

