
Concept explainers
Draw three resonance structures for the hydrogen sulfite ion (), one that obeys the octet rule for the central atom and two that expand the octet of the central atom. Calculate the formal charges on all atoms in each structure and determine which, if airy, of the resonance structures has formal charges that are inconsistent with the elements’ electronegativities.
Interpretation:
The resonance structures for hydrogen sulfite ion should be drawn where one resonance structure must obey octet rule and the other two having expanded octet. Also formal charge of all the resonance structure should be drawn and to identify the resonance structure that has an inconsistent with the electronegativities of the elements.
Concept Introduction:
- Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance.
- In some molecules, there is possibility of more than one Lewis structure where all the structures are equally acceptable. One of the acceptable Lewis structures of these molecules is called resonance structure. All the possible resonance structures are imaginary whereas the resonance hybrid is real.
- These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
- Atoms can be stable even though the number of valence electrons in the atoms in a molecule is more than 8 and is called expanded octet
To identify: the resonance structure of hydrogen sulfite ion
Answer to Problem 10PPA
Explanation of Solution
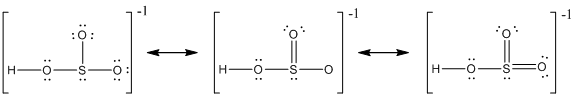
The resonance structure of
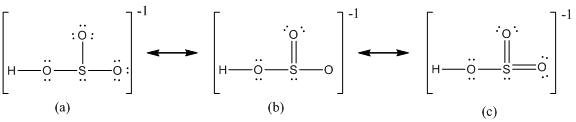
Delocalization of electrons takes place because of the presence of lone pair of electrons in the terminal atoms and the possibility of double bond. The chemical bonding of a molecule cannot be represented using a single Lewis structure and represented by structures (a), (b) and (c). The chemical bonding are described by delocalization of electrons forming three possible resonance structures. In all the resonance structures the position, over whole charge and chemical framework remains intact. Also in these structures only in the arrangement of the electrons differs not the relative position of the atomic nuclei.
Here sulfur shares a double bond with one of the oxygen atom and shares two single bond with remaining two oxygen atoms. Hence in (a), the central sulfur atom is surrounded by 8 electrons therefore it obeys octet rule.
Here sulfur shares a double bond with one of the oxygen atom and shares two single bond with remaining two oxygen atoms. It also have two lone pairs on it. Hence in (b), the central sulfur atom is surrounded by 10 electrons therefore it obeys does not octet rule and is an expanded octet
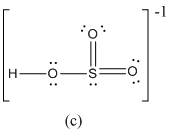
Here sulfur shares two double bond with oxygen atoms and shares a single bond with remaining one oxygen atoms. It also have two lone pairs on it. Hence in (c), the central sulfur atom is surrounded by 12 electrons therefore it obeys does not octet rule and is an expanded octet
Interpretation: the formal charge of atoms of the resonance structure of hydrogen sulfite ion should be determined.
Concept Introduction:
- A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
- This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
- Formal charge of an atom can be determined by the given formula.
To determine: the formal charge of atoms of the resonance structure of hydrogen sulfite ion.
Answer to Problem 10PPA
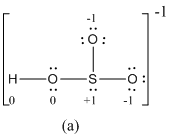
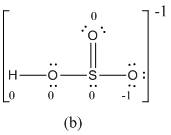
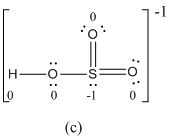
Explanation of Solution
For a
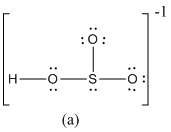
The formal charge of atoms in (a) is calculated.


- Sulfur atom
Substituting,
- Terminal oxygen atoms that has single bond with sulfur atom
Substituting,
- Oxygen atoms that has single bond with sulfur atom and hydrogen
Substituting,
- Hydrogen atom
Substituting,
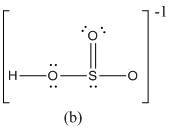
For b
The formal charge of atoms in (b) is calculated.


- Sulfur atom
Substituting,
- Terminal oxygen atom that has single bond with sulfur atom
Substituting,
- Terminal oxygen atom that has double bond with sulfur atom
Substituting,
- Oxygen atoms that has single bond with sulfur atom and hydrogen
Substituting,
- Hydrogen atom
Substituting,
For c
The formal charge of atoms in (c) is calculated.


- Sulfur atom
Substituting,
- Terminal oxygen atoms that has double bond with sulfur atom
Substituting,
Oxygen atoms that has single bond with sulfur atom and hydrogen
Substituting,
Hydrogen atom
Substituting,
- The electronegativity of oxygen is higher than sulfur so usually more electronegative receives negative formal charge. But in the case of resonance structure (c), the less electronegative sulfur atom has -1 formal charge where oxygen gets zero formal charge. Therefore resonance structure (c) has formal charges that are inconsistent with the electronegativities of the elements.
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: Atoms First
- the formal charges on all the atoms in the following Lewis diagrams. Which one would best represent bonding in the molecule Cl2O ?arrow_forwardThe Lewis structure of acetone is Circling the carbonyl carbon, i.e., the carbon atom attached to oxygen, and its octet gives Circling the oxygen atom and its octet gives Thus, atoms share electrons in making bonds, and a pair of electrons may be included in the octet of two different atoms. When computing the formal charge on an atom, the number of electrons that belong to that atom is compared with the number of electrons the atom would have in the unbonded and neutral state. If the two numbers are the same, the formal charge on the atom is zero. In a Lewis structure both electrons in an unshared pair belong to the atom, and one of every pair of shared (bonding) electrons belongs to the atom.arrow_forwardDraw resonance structures for each of these ions: NSO− and SNO−. (The atoms are bonded in the order given in each case, that is, S is the central atom in NSO−.) Use formal charges to determine which ion is likely to be more stable. Explain why the two ions cannot be considered resonance structures of each other.arrow_forward
- Two Lewis structures can be written for nitrosyl fluoride, which contains one nitrogen, one oxygen, and one fluorine atom per molecule. Write the two Lewis structures land assign a formal charge to each atom.arrow_forwardWrite the Lewis structure for nitrosyl fluoride, FNO. Using only a periodic table, identify (a) which is the longer bond. (b) which is the stronger bond. (c) which is the more polar bond.arrow_forwardChloromethane has the Lewis structure _______________________________ The carbon atom is sharing 4 electron pairs. In each shared pair the carbon atom “owns” 1 electron. The number of electrons that “belong” to carbon is ___. Carbon, being a Group ___ element would have 4 , outer shell electrons in the unbonded, neutral state. Therefore, the carbon atom in chloromethane has a formal charge of zero.arrow_forward
- Write all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forwardA complete Lewis structure must show all nonzero formal charges. Complete each of thefollowing Lewis structures by adding any missing formal charges.arrow_forwardThe cyanate ion (OCN- ) and the fulminate ion (CNO- ) share the same three atoms but have vastly different properties. The cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. The resonance structures of the cyanate ion are explored in Example 9.8. Draw Lewis structures for the fulminate ion—including possible resonance forms— and use formal charge to explain why the fulminate ion is less stable (and therefore more reactive) than the cyanate ion.arrow_forward
- Using only single bonds, draw two corect Lewis structures of HCIO, one with Cl as the central atom and the other with O as the central atom. Calculate the formal charges of all atoms in each molecule. State which is the more likely structure based on the formal charges. In the previous problem, you drew two structures of HCIO. Are these resonance structures? Explain why or why not.arrow_forwardWhat determines which atoms are the central atoms? How do you adjust the number of electrons to form a cation? What about an anion? What causes some molecules with the same number of electron domains to have different shapes? Would they cause the real bond angle to be larger or smaller? Why? Which of the following elements should you expect to form an incomplete octet: H, He, Be, B, C, O? Why? Which of the following elements are good candidates to form an expanded octet: N, F, Ne, P, S, Cl, Ar? Why? What things must you consider when determining if a molecule is polar or non-polar?arrow_forwardDraw two different Lewis structures for ClO3-, one which all atoms follow the octet rule and one where the chlorine has an expanded octet. Determine the formal charge on all atoms for each structure. Which do you think is the better Lewis structure? Explain your reasoning.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





