
Concept explainers
Draw Lewis structures for the following four isoelectronic species: (a) CO, (b) NO+, (c) CN−, (d) N2. Show formal charges. (See Problem 6.69.)
(a)
Interpretation: The Lewis structures for the given isoelectronic species should be shown.
Concept Introduction:
- Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
- It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
To find: The Lewis structure for the given set of isoelectronic species.
Answer to Problem 6.83QP

Explanation of Solution
Given isoelectronic species is below.
Lewis structure of the above isoelectronic species is drawn below.

The total number of valence electrons is found to be 10, where carbon and oxygen has 4 and 6 valence electrons respectively.
The 8 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on nitrogen atom to complete the octet.
Since the octets of carbon atoms are not filled, a triple bond was made between carbon and oxygen atoms in expense of two electrons where the remaining four electrons are distributed equally over two atoms present in the given species.
(b)
Interpretation: The Lewis structures and the formal charges for the given isoelectronic species should be shown.
Concept Introduction:
- Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
- It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
To find: The Lewis structure for the given set of isoelectronic species.
Answer to Problem 6.83QP
Explanation of Solution
Given isoelectronic species is below.
Lewis structure of the above isoelectronic species is drawn below.

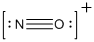
The total number of valence electrons is found to be 11, where nitrogen and oxygen contains 5 and 6 valence electrons respectively. The whole charge of the molecule is +1 that results in the total number of valence electrons as 10.
The 8 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on nitrogen atom to complete the octet.
Since the octets of nitrogen atoms are not filled, a triple bond is made between nitrogen and oxygen atoms in expense of two electrons where the remaining four electrons are distributed over the 2 atoms present in the given molecule.
(c)
Interpretation: The Lewis structures and the formal charges for the given isoelectronic species should be shown.
Concept Introduction:
- Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
- It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
To find: The Lewis structure for the given set of isoelectronic species.
Answer to Problem 6.83QP

Explanation of Solution
Given isoelectronic molecule is below.
Lewis structure of the above isoelectronic species is drawn below.

The total number of valence electrons is found to be 9, where nitrogen and carbon contributes 5 and 4 electrons respectively. The whole charge of the molecule is -1 making the total number of valence electrons 10.
The 8 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on nitrogen atom to complete the octet.
Since the octets of carbon atoms are not filled, a triple bond is made between carbon and nitrogen atoms in expense of two electrons where the remaining four electrons are distributed over the atoms present in the given molecule.
(d)
Interpretation: The Lewis structures and the formal charges for the given isoelectronic species should be shown.
Concept Introduction:
- Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
- It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
To find: The Lewis structure for the given set of isoelectronic species.
Answer to Problem 6.83QP

Explanation of Solution
Given isoelectronic species is below.
Lewis structure of above isoelectronic species is drawn below.

The total number of valence electrons is found to be 10, where both nitrogen atoms contribute 5 electrons.
The 8 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on nitrogen atom to complete the octet.
Since the octets of nitrogen atoms are not filled, a triple bond is made between both nitrogen atoms.
(a)
Interpretation: The formal charges for the given isoelectronic species should be shown.
Concept Introduction
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
The Lewis structure with formal charge on each of the atoms close to zero is taken as the most plausible structure.
Formal charge of an atom can be determined by the given formula.
Answer to Problem 6.83QP
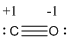
Explanation of Solution
Formal charge of the given isoelectronic species is given below
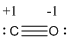
The formal charge of the given isoelectronic species is calculated,
- Carbon atom
Substituting these values to the equation,
- Oxygen atom
Substituting these values to the equation,
(b)
Interpretation: The formal charges for the given isoelectronic species should be shown.
Concept Introduction
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
The Lewis structure with formal charge on each of the atoms close to zero is taken as the most plausible structure.
Formal charge of an atom can be determined by the given formula.
Answer to Problem 6.83QP
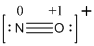
Explanation of Solution
Formal charge of the given isoelectronic species is given below
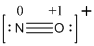
The formal charge of the given isoelectronic species is calculated,
- Nitrogen atom
Substituting these values to the equation,
- Oxygen atom
Substituting these values to the equation,
(c)
Interpretation: The formal charges for the given isoelectronic species should be shown.
Concept Introduction
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
The Lewis structure with formal charge on each of the atoms close to zero is taken as the most plausible structure.
Formal charge of an atom can be determined by the given formula.
Answer to Problem 6.83QP
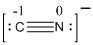
Explanation of Solution
Formal charge of the given isoelectronic species is given below
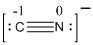
The formal charge of the given isoelectronic species is calculated,
- Carbon atom
Substituting these values to the equation,
- Nitrogen atom
Substituting these values to the equation,
(d)
Interpretation: The formal charges for the given isoelectronic species should be shown.
Concept Introduction
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
The Lewis structure with formal charge on each of the atoms close to zero is taken as the most plausible structure.
Formal charge of an atom can be determined by the given formula.
Answer to Problem 6.83QP
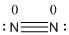
Explanation of Solution
Formal charge of the given isoelectronic species is given below
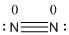
The formal charge of the given isoelectronic species is calculated,
- Nitrogen atom
Substituting these values to the equation,
- Since both the nitrogen atoms are similar, the formal charge of the nitrogen atoms is zero.
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: Atoms First
- Use Lewis electron-dot symbols to represent the formationof (a) BrF₃ from bromine and fluorine atoms; (b) AlF₃ from alu-minum and fluorine atoms.arrow_forward(c) Draw the orbital diagrams and Lewis symbols to depict the formation of Na* and CI ions from the atoms. Give the formula of the compound formed. (d) The predicted bond length for HF is 109 pm (the sum of the covalent radii of H, 37 pm and F. 72 pm), however the actual bond length for HF is shorter (92 pm). It was observed that the difference between predicted and actual bond lengths becomes smalleor going down the halogen group from HF to HI Describe these observationsarrow_forward8.4 Write Lewis dot symbols for the following ions: (a) Li+,(b) Cl−, (c) S2−, (d) Sr2+, (e) N3−.arrow_forward
- Draw a Lewis electron-dot symbol for (a) Ba; (b) Kr; (c) Br.arrow_forward8D.4. Benzene, a common solvent, is a covalent molecular compound which contains only carbon and hydrogen. Its simplest (empirical) formula is CH, and its molecular weight is 78 g/mol to 2 significant digits. What is its molecular formula? 8D.5. Draw the Lewis Dot Structure of (a) phosphorus pentafluoride. What is the formal charge on all atoms? (b) the perchlorate ion- in this case expand the octet for the chlorine atom in order to optimize the formal charges.arrow_forward8.23 Use Lewis dot symbols to show the transfer of electronsbetween the following atoms to form cations andanions: (a) Na and F, (b) K and S, (c) Ba and O,(d) Al and N.arrow_forward
- Write the Lewis symbol for atoms of each of the following elements:(a) Al, (b) Br, (c) Ar, (d) Sr.arrow_forwardIn the vapor phase, BeCl2 exists as a discrete molecule. (a) Draw the Lewis structure of this molecule, using only single bonds. Does this Lewis structure satisfy the octet rule? (b) What other resonance structures are possible that satisfy the octet rule? (c) On the basis of the formal charges, which Lewis structure is expected to be dominant for BeCl2?arrow_forwardOne of the following pictures(Figure 1) represents NaCl and one represents MgO. Which is which? (a) is NaCl and (b) is MgO (b) is NaCl and (a) is MgO Which has the larger lattice energy? NaCl MgOarrow_forward
- Consider the formate ion, HCO2", which is the anion formed when formic acid loses an H* ion. The H and the two O atoms are bonded to the central C atom. (a) Draw the best Lewis structure(s) for this ion. (b) Are resonance structures needed to describe the structure? Explain briefly (c) Would you predict that the C-O bond lengths in the formate ion would be longer or shorter relative to those in CO2? Explain brieflyarrow_forwardAn ionic bond is formed between a cation A+and an anion B−. How would the energy of theionic bond [see Equation (9.2) of your textbook] be affected by the following changes?(a) doubling the radius of A+,(b) tripling the charge on A+,(c) doubling the charges on A+and B−,(d) decreasing the radii of A+and B−to half their original values.[1 point each = 4 points]arrow_forwardConsider the following compounds: CaCl 2 , CaI 2 , and MgCl 2 . Answer the following questions based on expected periodic trends: (a) Which is expected to have t he shortest ionic bonds? (b) Which is expected to have the highest lattice energy? (c) Which is expected to have the lowest melting point?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





