
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 4.6, Problem 14P
Interpretation Introduction
Interpretation:
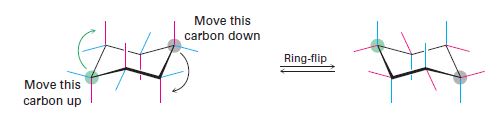
The coloured–green, red or blue colour bond is to be identified in cyclohexane in the starting and the flipped structure.
Concept introduction:
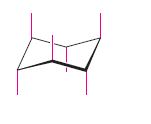
Axial bond in cyclohexane
Equatorial bond in cyclohexane
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Shown are two stereo representations of lactic acid. Compare the two structures shown. Determine whether they
represent identical molecules or mirror images.
identical
COOH
COOH
H"
OH
CH3
H3C
HO
Determine the absolute configuration of the two structures.
Left: S✔ Right: S
An error has been detected in your answer. Check for typos,
miscalculations etc. before submitting your answer.
H
List the name and position for each substituent in this organic molecule on a separate row of the table below:
Your answer for the name column should be just one word. In the position column, write the number of the carbon where
the substituent is attached to the parent ring (which has been highlighted for you).
Note for advanced students: You may assume that the parent ring is numbered in a counter-clockwise fashion with position
#1 at the top of this molecule.
You can add more rows to the table if you need them.
Add Row
name
B
position
0
G
What is the absolute configuration at C1 and C2?
Chapter 4 Solutions
Organic Chemistry
Ch. 4.1 - Give IUPAC names for the following cycloalkanes:Ch. 4.1 - Draw structures corresponding to the following...Ch. 4.1 - Name the following cycloalkane:Ch. 4.2 - Prob. 4PCh. 4.2 - Draw the structures of the following molecules:...Ch. 4.2 - Prostaglandin F2α, a hormone that causes uterine...Ch. 4.2 - Name the following substances, including the cis-...Ch. 4.3 - Each H↔H eclipsing interaction in ethane costs...Ch. 4.3 - cis-1, 2-Dimethylcyclopropane has more strain than...Ch. 4.4 - Prob. 10P
Ch. 4.4 - Two conformations of cis-l, 3-dimethylcyclobutane...Ch. 4.6 - Draw two different chair conformations of...Ch. 4.6 - Draw two differant chair conformations of trans-1,...Ch. 4.6 - Prob. 14PCh. 4.7 - What is the energy difference between the axial...Ch. 4.7 - Prob. 16PCh. 4.7 - Look at Figure 4-12 on page 105, and estimate the...Ch. 4.8 - Draw the more stable chair conformation of the...Ch. 4.8 - Identify each substituent in the following...Ch. 4.9 - Which isomer is more stable, cis-decalin or...Ch. 4.9 - Look at the following structure of the female...Ch. 4.SE - Prob. 22VCCh. 4.SE - Name the following compound, identify each...Ch. 4.SE - A trisubstituted cyclohexane with three...Ch. 4.SE - The following cyclohexane derivative has three...Ch. 4.SE - Prob. 26VCCh. 4.SE - Draw the five cycloalkanes with the formula C5H10.Ch. 4.SE - Draw two constitutional isomers of cis-1,...Ch. 4.SE - Prob. 29APCh. 4.SE - Tell whether the following pairs of compounds are...Ch. 4.SE - Prob. 31APCh. 4.SE - Prob. 32APCh. 4.SE - Draw 1, 3, 5-trimethylcyclohexane using a hexagon...Ch. 4.SE - Hydrocortisone, a naturally occurring hormone...Ch. 4.SE - A 1, 2-cis disubstituted cyclohexane, such as...Ch. 4.SE - A 1, 2-trans disubstituted cyclohexane must have...Ch. 4.SE - Prob. 37APCh. 4.SE - Which is more stable, a 1, 4-trans disubstituted...Ch. 4.SE - cis-1, 2-Dimethylcyclobutane is less stable than...Ch. 4.SE - From the data in Figure 4-12 and Table 4-1,...Ch. 4.SE - Prob. 41APCh. 4.SE - Draw the two chair conformations of...Ch. 4.SE - Draw the two chair conformations of...Ch. 4.SE - Galactose, a sugar related to glucose, contains a...Ch. 4.SE - There are four cis-trans isomers of menthol...Ch. 4.SE - There are four cis-trans isomers of menthol...Ch. 4.SE - The diaxial conformation of cis-1,...Ch. 4.SE - Approximately how much steric strain does the...Ch. 4.SE - In light of your answer to Problem 4-43, draw the...Ch. 4.SE - Prob. 50APCh. 4.SE - Prob. 51APCh. 4.SE - Using molecular models as well as structural...Ch. 4.SE - trans-Decalin is more stable than its cis isomer,...Ch. 4.SE - As mentioned in Problem 3-53, the statin drugs,...Ch. 4.SE - myo-Inositol, one of the isomers of...Ch. 4.SE - How many cis–trans stereoisomers of myo-inositol...Ch. 4.SE - The German chemist J. Bredt proposed in 1935 that...Ch. 4.SE - Tell whether each of the following substituents on...Ch. 4.SE - Prob. 59APCh. 4.SE - Prob. 60APCh. 4.SE - Ketones react with alcohols to yield products...Ch. 4.SE - Alcohols undergo an oxidation reaction to yield...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Construct a model in which a tetrahedral carbon atom has four different colored model atoms attached to it- red, green, orange and white representing 4 different atoms attached to the central atom. a) Does the atom have a plane of symmetry? why or why not? b) Now replace the green atom in your model with a second orange atom. Now two of the groups attached to the carbon atom are identical. Does the model now have a plane of symmetry? Describe it. c)A carbon atom has four different groups attached to the stereogenic center. Draw structural formulas for the following compound and mark stereogenic centers with as asterisk: 1-bromobutane, 2-bromobutane, 1,2-dibromobutane, 1,4-dibromobutane, 2,3-dibromobutane.arrow_forwardDraw the chair flip of the following molecule. DO NOT rotate the molecule in anyway. Et Me After the chair flip, which letter corresponds to the position of the methyl group? After the chair flip, which letter corresponds to the position of the ethyl group? BC A K D L E F G Harrow_forwardHighlight each chiral center in the following molecule. If there are none, then check the box under the drawing area. There are no chiral centers. ||| OH Xarrow_forward
- Which of these is the correct rotation for [a] of 0°?arrow_forwardLook for an axis of rotation in this molecule. Make sure to rotate the molecule to see all of it. C-Carbon H-Hydrogen F-Fluorine Check the correct answer. There is one axis and the molecule can be rotated 120°. There is no symmetry axis. There are four axes and the molecule can be rotated 120°. There is one axis and the molecule can be rotated 90°. 9 Rotate X 9 Rotate Y 5 Rotate Z Zoom In Q Zoom Out A Label Atomsarrow_forwardHighlight each chiral center in the following molecule. If there are none, then check the box under the drawing area. There are no chiral centers. Xarrow_forward
- Construct the three-dimensional model of 2-methylhexane, also draw the anti and gauche models where the rotation between carbons 3 and 4 is clearly appreciated.arrow_forwarddumb question. Theres a C≡C in the name of the compound, but theres a note that says no C≡C visible. Are there instances when theres a C≡C in the compound, even tho it doesnt show up on the IR? just worried how to answer a test question asking about a compound and if there will be a C≡C in it even if its not seenarrow_forwardCheck the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of the above box under the table. * Molecule 1 Molecule 4 q Onone of the above Molecule 2 Molecule 5 Molecule 3 Molecule 6 *** X 0arrow_forward
- a) Draw one isomer of C6H14. b) Draw one isomer of C6H12- c) Draw one isomer of C6H140 that exhibits hydrogen bonding. d) Draw one isomer of C6H140 that is not capable of hydrogen bonding. BONUS: Show all locations of possible hydrogen bonding for the C6H140 isomer that you drew above in part c.arrow_forwardQUESTION 3 In the answer boxes, indicate whether carbon 2 compound. CI G CI ||Z and carbon 3 have R or 8 configuration in the followingarrow_forwardQUESTION 1 When calibrating the polarimeter the angle of rotation should be at 0 degrees. Another indication of a calibrated i when polarimeter is when one of the semicircle halves is bright and the ther is dim. Which of the staetement is correct when calibrating a polarimeter? O Only the first statement is correct O Only the second statement is correct both statements are correct none of the statements is correct.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY