
Concept explainers
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe, and the temperature of the CO2 drops to 450 K at the exit. Determine the volume flow rate and the density of carbon dioxide at the inlet and the volume flow rate at the exit of the pipe using (a) the ideal-gas equation and (b) the generalized compressibility chart. Also, determine (c) the error involved in the first case.
FIGURE P3–89
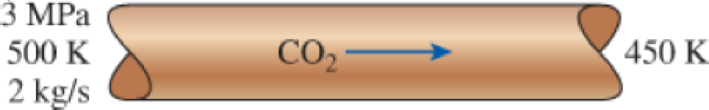
(a)
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the ideal gas equation of state.
Answer to Problem 89P
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the ideal gas equation of state are
Explanation of Solution
Refer to Table A-1, obtain the gas constant, critical pressure, and the critical temperature of carbon dioxide.
Write the equation of volume flow rate at the inlet of the pipe.
Here, inlet temperature and inlet pressure are
Calculate the density at the inlet of pipe.
Calculate the equation of volume flow rate at the outlet of the pipe.
Here, outlet temperature and outlet pressure are
Conclusion:
Substitute
Substitute
Substitute
Thus, the volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the ideal gas equation of state are
(b)
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the generalized compressibility chart.
Answer to Problem 89P
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the generalized compressibility chart are
Explanation of Solution
Calculate the equation of reduced pressure at the inlet of the pipe.
Here, the critical pressure is
Calculate the equation of reduced temperature at the inlet of the pipe.
Here, the critical temperature is
Calculate the equation of reduced pressure at the outlet of the pipe.
Calculate the equation of reduced temperature at the outlet of the pipe.
Write the equation of volume flow rate at the inlet of the pipe.
Here, compressibility factor at the inlet of pipe is
Calculate the density at the inlet of pipe.
Calculate the equation of volume flow rate at the outlet of the pipe.
Here, compressibility factor at the outlet of pipe is
Conclusion:
Substitute 3 MPa for
Substitute 500 K for
Substitute 3 MPa for
Substitute 450 K for
Refer to Figure 3-48, obtain the compressibility factor at inlet state
Refer to Figure 3-48, obtain the compressibility factor at outlet state
Substitute 0.9791 for
Substitute 0.9791 for
Substitute 0.9656 for
Thus, the volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the generalized compressibility chart are
(c)
The error involved in the first case.
Answer to Problem 89P
The error involved in the first case are
Explanation of Solution
Calculate the percentage of error involved in the first case of volume flow rate at the inlet condition.
Here, calculated volume flow rate at inlet state from EOS is
Calculate the percentage of error involved in the first case of density at the inlet condition.
Here, calculated density at inlet state from EOS is
Calculate the percentage of error involved in the first case of volume flow rate at the outlet condition.
Here, calculated volume flow rate at outlet state from EOS is
Conclusion:
Substitute
Substitute
Substitute
Thus, the error involved in the first case are
Want to see more full solutions like this?
Chapter 3 Solutions
Thermodynamics: An Engineering Approach
- Determine the volume flow rate and the density of carbon dioxide at the inlet and the volume flow rate at the exit of the pipe using (a) the ideal-gas equation and (b) the generalized compressibility chart. Also, determine (c) the error involved in the first case.arrow_forwardA rigid container with a volume of 2000 liters contains 4 kg mixture of saturated water and steam at 120°C. The mixture is slowly heated until the liquid content is completely vaporized. Determine the following: a) quality of the vapor at the initial condition; and b) temperature of water after heating. From steam tables @t: = 120°C (vn = 1.0603x10' m'/kg, vạ1 = 891.9x10' m²/kg). HINT: If v is 508.9 x10 there are 140°C, if v = 495.6x10 there are 141°carrow_forwardThe piston in a car engine compresses a mixture of gasoline vapour and air from 700ml to 100ml during its compression stroke. Before the compression stroke, the mixture had a pressure of 75 kPa and a temperature of 120celcius. What is the pressure of the cylinder after the compression stroke if the temperature of the gas rises to 200celcius? Express your answer in MPa with two significant digits.arrow_forward
- Determine the specific volume of refrigerant-134a at 1 MPa and 50°C, using (a) the ideal-gas equation of state and (b) the generalized compressibility chart. Compare the values obtained to the actual value of 0.021796 m3/kg and determine the error involved in each case.arrow_forwardA piston-cylinder device is filled with 2.75 kg of carbon monoxide (CO). The inital volume and pressure of the gas are 0.05 m3 and 250 kPa, respectively. The piston is forced to move so that the gas isothermally expands three times of its original volume. Estimate the final pressure of the gas by stating all your assumptions clearly.arrow_forwardA 200 liter tank contains a mixture of saturated liquid water and saturated steam at 370°C. Find the mass of each phase if their volumes are equal and the colume iccupied by each phase if their masses are equal. Given vf=2.213x10-3m3/kg and vg=4.925x10-3 m3/kgarrow_forward
- Saturated water vapor at 200.111 oC is isothermally condensed to a saturated liquid in a piston-cylinder device. Calculate (a) the heat transfer (b) change of internal energy, (c) the work done during the process, (d) change of entropy, and (e) change of enthalpy. Show T-s diagramarrow_forwardNitrogen (N2) at 100C and 600 kPa expands in such a way that it can be approximated by a polytropic process with polytropic index n = 1.2. Calculate the work and the heat transfer per kg of N2 if the final pressure is 100 kPa. Draw this expansion process on P- v diagram. For nitrogen, take the gas constant R = 0.297 kJ/(kgK) and cv = 0.745 kJ/(kgK). [Hint: Use the non-flow energy equation, NFE E.].arrow_forwardNitrogen (N2) at 100C and 600 kPa expands in such a way that it can be approximated by a polytropic process with polytropic index n = 1.2. Calculate the work and the heat transfer per kg of N2 if the final pressure is 100 kPa. Draw this expansion process on P- v diagram. For nitrogen, take the gas constant R = 0.297 kJ/(kgK) and cv = 0.745 kJ/(kg K).arrow_forward
- Carbon dioxide of mass 0.9 kg is contained in a vessel at a pressure of 4 MPa and atemperature of 500 °C. The gas is allowed to expand into an empty connected vessel, duringwhich its temperature falls to 120 °C. The combined volume of the two vessels is 0.45 m3.Determine (i) the initial volume of the carbon dioxide, (ii) its final pressure and (iii) thepolytropic index ‘n’ of the expansion process. Carbon dioxide has a molecular weight of 44 and the universal gas constant is 8314 Jkmol-1K-1.arrow_forwardA piston-cylinder device initially contains 0.4 m^3 of saturated water vapor at a pressure of 200 kPa. At this time, a linear spring (K = 150 N/m, no other information about spring provided) is touching the piston, but exerts no force upon it. Heat is now transferred to the water vapor for 30 minutes through a resistive heater having a current of 10 amperes. The pressure and volume rise to 300 kPa and 0.6 m^3 while the system lost 10 kJ of heat during this process. Determine the voltage of the heater. Show detailed calculations and specify any required values found on thermodynamic tables.arrow_forwardWhat is the change in the enthalpy, in kJ/kg, of oxygen as its temperature changes from 150 to 250°C? The constant-pressure specific heat of oxygen at room temperature is cp = 0.918 kJ/kg-K. Is there any difference if the temperature were to change from 0 to 100°C? Consider the variation of specific heat with temperature. Cp= 0.964 kJ/kg-K at 200°C and cp= 0.934 kJ/kg-K at 100°C. The change in the enthalpy of oxygen is kJ/kg. The difference between the first case and the second case is %.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





