
Concept explainers
The spring-loaded piston–cylinder device shown in Fig. P3–63 is filled with 0.5 kg of water vapor that is initially at 4 MPa and 400°C. Initially, the spring exerts no force against the piston. The spring constant in the spring force relation F = kx is k = 0.9 kN/cm and the piston diameter is D = 20 cm. The water now undergoes a process until its volume is one-half of the original volume. Calculate the final temperature and the specific enthalpy of the water.
FIGURE P3–63
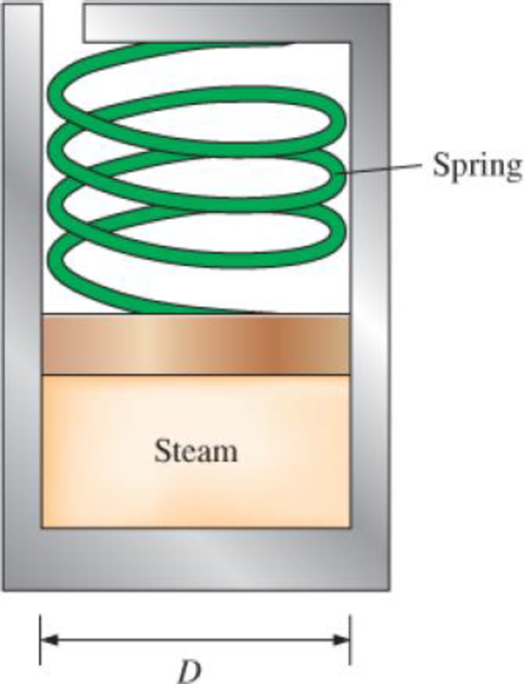
The final temperature of the spring-loaded piston-cylinder device.
The enthalpy of the spring-loaded piston-cylinder device.
Answer to Problem 63P
The final temperature of the spring-loaded piston-cylinder device is
The enthalpy of the spring-loaded piston-cylinder device is
Explanation of Solution
Write the final temperature of the spring-loaded piston-cylinder device using linear
Here, the spring constant is
Determine the final specific volume of the spring-loaded piston-cylinder device.
Determine the quality of final state for the spring-loaded piston-cylinder device.
Here, the specific volume of saturated liquid is
Determine the enthalpy at the final state of the spring-loaded piston-cylinder device.
Here, the specific enthalpy of saturated liquid is
Conclusion:
From the Table A-6, “Superheated water” to obtain the value of the specific volume of steam at 4 MPa of pressure and
Substitute
Substitute
Refer to Table A-6, “Saturated water”, obtain the below properties at the final pressure
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y are pressure and temperature.
Show the pressure at
| S. No |
Pressure, kPa |
Temperature, |
| 1 | 218.41 | |
| 2 | ||
| 3 | 223.95 |
Calculate final temperature at the pressure
Substitute
From above calculation the final temperature at the pressure
Thus, the final temperature of the spring-loaded piston-cylinder device is
Repeat the above Equation (V), to obtain the value of specific volume of saturated liquid, the specific volume of saturated vapour, specific enthalpy of saturated liquid and the specific enthalpy of saturated vapour at the final pressure
Substitute
Substitute
Thus, the enthalpy of the spring-loaded piston-cylinder device is
Want to see more full solutions like this?
Chapter 3 Solutions
Thermodynamics: An Engineering Approach
- A piston–cylinder device contains 0.05 m3 of a gas initially at 200 kPa. At this state, a linear spring that has a spring constant of 150 kN/m is touching the piston but exerting no force on it. Now heat is transferred to the gas, causing the piston to rise and to compress the spring until the volume inside the cylinder doubles. If the crosssectional area of the piston is 0.25 m2 , determine (a) the final pressure inside the cylinder, (b) the total work done by the gas, and (c) the fraction of this work done against the spring to compress it.arrow_forwardA piston-cylinder assembly contains air at 210 kPa and occupies a volume of 0.01m. The piston is attached to one end of a spring and the other end of the spring is fixed to a wall. The force exerted by the spring on the piston is proportional to the decrease in the length of the spring. The ambient atmospheric pressure is 100 kPa. Now, the air in the cylinder is heated till the volume is doubled and at this instant it is found that the pressure of the air in the cylinder is 600 kPa. Sketch the process on p-v diagram and calculate work done by the gas. Q2:arrow_forwardA piston-cylinder device contains 0.8 kg of steam at 300 0C and 1 MPa. Steam is cooled at constant pressure until one-half of the mass condenses. (a) Find the final temperature. (b) Determine the volume change.arrow_forward
- A weighted piston-cylinder device is initially filled with water as a saturated vapor at 500 kPa. The device is then heated isobarically until the temperature is 360 °C. Determine the heat transfer per unit mass, in kJ/kg. Sketch the process on a T-V diagram and submit with your work.arrow_forwardThe figure shown above is a 0.5 cm², 5 kg cylinder with a moving piston attached to a spring. The cylinder is filled with unknown gas and initially the spring did not exert any force. Determine the work done when the spring is compressed by 5cm. Assume initial volume is 0.005m³ and k= 100 N/m. Neglect the effect of heat and internal energy.arrow_forward1- A piston-cylinder device whose piston is resting on top of stops initially contains 0.5 kg of helium gas at 100 kPa and 25 C. The mass of the piston is such that 500 kPa of pressure is required to raise it. (1) How much heat must be transferred to the helium before the piston starts rising and (2) show the process on P-V diagramarrow_forward
- The piston cylinder system shown below contains R-410a at 20°C and 100 kPa. If the temperature of the R-410a is increased to 50°C in a constant pressure process, what is the specific volume of the R-410a in the cylinder at the final state?arrow_forwardA piston/cylinder arrangement contains 2 kg of ammonia at a temperature 132.45 ℃ and a pressure 5 MPa. The piston floats. Heat is added to the system until the temperature of ammonia reaches 416.37 ℃. (a) Determine the phase of ammonia during the whole process (from the initial to the final state). (b) Show the initial and final points of the process in a T-s and p-v diagram. In both diagrams sketch the dome. Show these two points relative to the dome. (c) Determine the initial and final volume of the piston/cylinder arrangement using tables.arrow_forwardA piston/cylinder arrangement contains 2 kg of ammonia at a temperature 132.45 ℃ and a pressure 5 MPa. The piston floats. Heat is added to the system until the temperature of ammonia reaches 416.37 ℃. (a) Determine the phase of ammonia during the whole process (from the initial to the final state). (b) Show the initial and final points of the process in a T-s and p-v diagram. In both diagrams sketch the dome. Show these two points relative to the dome.arrow_forward
- 3-39 Water initially at 200 kPa and 300°C is contained in a piston-cylinder device fitted with stops. The water is allowed to cool at constant pressure until it exists as a saturated vapor and the piston rests on the stops. Then the water continues to cool until the pressure is 100 kPa. On the T-v diagram, sketch, with respect to the saturation lines, the process curves pass- 1stams! od on TURNACE ing through the initial, intermediate, and final states of the 1992 palam 11. water. Label the T, P, and U values for end states on the process artita ne odrow OFTİ 21 nucfortyd beinvarian curves. Find the overall change in internal energy between the 1610 PRICE initial and final states per unit mass of water. al state Onimals uliod Anub 1516w 3-150 3-160 In Water 200 kPa 300°C 3-17C ment rises. Now on top of air Obs will rise in a co FIGURE P3-39 180 Qarrow_forwardNitrogen (N2) at 100C and 600 kPa expands in such a way that it can be approximated by a polytropic process with polytropic index n = 1.2. Calculate the work and the heat transfer per kg of N2 if the final pressure is 100 kPa. Draw this expansion process on P- v diagram. For nitrogen, take the gas constant R = 0.297 kJ/(kgK) and cv = 0.745 kJ/(kgK). [Hint: Use the non-flow energy equation, NFE E.].arrow_forwardA piston/cylinder assembly with a spring contains 0.075 kg of water. The piston is initially resting on the stops and the water has a temperature of T1 = 80°C and a quality of x1 = 30%. Heat is added to the water causing the piston to lift off the stops, rise up and compress the spring. Heating contin- ues until a final state is reached and the pressure is 200 kPa. The vertical distance between the top of the piston when it is resting on the stops and the bottom of the spring is Ay = 0.6 m, as shown in the figure. Assume the piston is massless (m, = 0), the area of the piston is A, = 0.12 m², the spring constant is k, = 40 kN/m and atmospheric pressure is Po = 100 kPa. a) Determine the volume, pressure and temperature of the water when the piston just lifts off the stops, V2, P2 and T2. b) Determine the pressure and specific volume of the water when the piston just touches but is not yet compressing the spring, P3 and v3. c) Determine the final volume of water in the piston/cylinder…arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





