
Concept explainers
Interpretation:
The structure of the productformed by intramolecular Diels–-Alder reaction of the given compound at room temperature is to be drawn.
Concept introduction:
>The conjugate addition of
Dienophile is the alkene that adds to diene.Diene is an electron-rich system attacksan electron deficient dienophile.The diene and dienophile, when residing in a single molecule, the intramolecular Diels–-Alder reaction is possible.
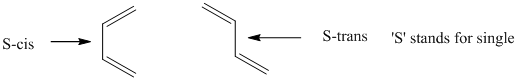
>In order to form the cyclic transition state in Diels–-Alder reaction, the conjugated diene must be able to adopt
The
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry - Standalone book
- When anthracene is added to the reaction of chlorobenzene with concentrated NaOH at 350 °C, an interesting Diels–Alderadduct of formula C20H14 results. The proton NMR spectrum of the product shows a singlet of area 2 around d 3 and abroad singlet of area 12 around d 7. Propose a structure for the product, and explain why one of the aromatic rings ofanthracene reacted as a dienearrow_forwardWhen heated, allyl aryl ethers and allyl vinyl ethers undergo a reaction called a Claisen rearrangement, a concerted reorganization of bonding electrons similar to the Diels-Alder reaction. The reaction proceeds through a six-membered, cyclic transition state. Draw the structure of the expected product when this compound undergoes a Claisen rearrangement. You do not have to consider stereochemistry. For the purposes of this problem, assume that double bonds in an aromatic ring are localized at the positions indicated in the figure. Include isomerization to a phenol IF appropriate.arrow_forwardHow could the following compounds be synthesized using a Diels–Alder reaction?arrow_forward
- One step in the synthesis of dodecahedrane (Section 4.11) involves reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4. This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.arrow_forward| Anthracene readily undergoes a Diels-Alder reaction with tetracyanoethene, even though anthracene is NC CN ? + aromatic. NC CN (a) Draw two possible products that can form from this reaction. (b) Explain why anthracene can readily undergo a Diels-Alder reaction, whereas benzene does not.arrow_forwardThe following triene undergoes an intramolecular Diels-Alder reaction to give a bicyclic product. Propose a structural formula for the product. Account for the observation that the Diels-Alder reaction given in this problem takes place under milder conditions (at lower temperature) than the analogous Diels-Alder reaction shown in Problem 20.34.arrow_forward
- Draw the products of the following Diels–Alder reactions. Indicate stereochemistry where appropriate.arrow_forwardThe following compound undergoes an intramolecular Diels-Alder reaction to give a tricyclic product. Propose a structural formula for the product. heat An intramolecular Diels-Alder adductarrow_forwardShow how the following starting materials are converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forward
- Indicate the reagents (diene and phyllodiene) necessary to obtain the following compound by a Dirls-Alder reaction. CH3 CH3 COOH COOHarrow_forward2) Rank the following dienes by how rapidly they will undergo Diel-Alder reactions with ethylene.arrow_forwardShow how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
