
Concept explainers
Write structural formulas for each of the following:
Interpretation:
The structural formula of the given compounds is to be written.
Concept introduction:
When writing the structural formula of any compound, first the functional group from the suffix of the given name is identified.
The longest carbon chain containing the functional group is located.
The carbon atoms of the chain are numbered in a way that the functional group is at lowest numbered carbon atom.
Substituents are attached to the parent chain according to their positions given in the name.
In alkenes, the Z isomers have the higher ranked substituents on the same side of the double bond, and in E isomers, higher ranked substituents are on the opposite sides of the double bond.
Answer to Problem 26P
Solution:
a)
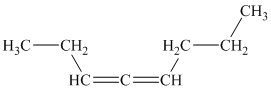
b)
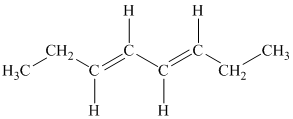
c)
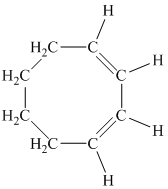
d)
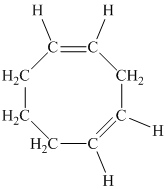
e)
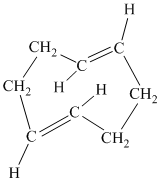
f)
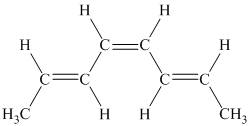
g)
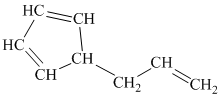
h)
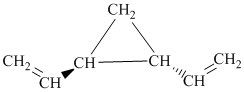
i)
Explanation of Solution
a) The given compound is
The parent name contains the word “
The structural formula is as follows:

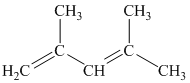
b) The given compound is
The parent name contains the word “
The structural formula is as follows:
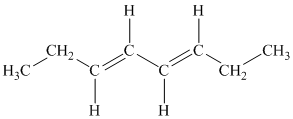
c) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
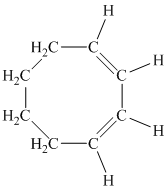
d) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
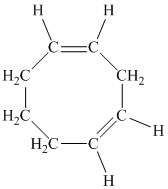
e) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
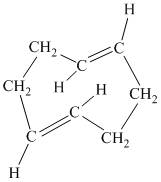
f) The given compound is
The parent name contains the word “
The structural formula is as follows:
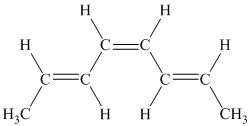
g) The given compound is
The parent name contains the word “cyclopenta” in it, which means that the parent carbon chain contains five carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
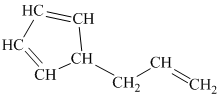
h) The given compound is
The parent name contains the word cyclopropane in it, which means that the parent carbon chain contains three carbon atoms in a cyclic form. There are two vinyl groups attached to the cyclopropane. The numbers
The structural formula is as follows:
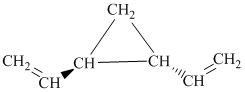
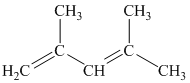
i) The given compound is
The parent name contains the word “penta” in it, which means that the longest carbon chain contains five carbon atoms. The suffix added is
The structural formula is as follows:
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry - Standalone book
- 12-77 Show how to convert cyclopentene into these compounds. 1,2-Dibromocyclopentane Cyclopentanol Iodocyclopentane Cyclopentanearrow_forwardAddition of HBr to 1,3-Butadiene produce A- 3-Bromo-1-butene B- 1-Bromo-2-butene C- 3,4-dibromo-1-butene D-A and B A O BO C Oarrow_forwardC. How many of the following structures are allylic? D. How many are arylic? E. How many are benzylic?arrow_forward
- Name the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H CH3 H3C C=C H2C=CHCHÇH H ČH3arrow_forwardBromine adds to cis- and trans-2-butene to give different diastereomers of 2,3-dibromo- butane. What does this say about the mode of addition of bromine to this alkene? H3C Br Bry H CH, Br `H. H,C Br Brg HH Br CH3arrow_forwardThe name of this compound. Br H. C=C Cl F O Z-2-bromo-2-chloro-1-fluoroethene E-1-bromo-1-chloro-2-fluoroethene O cis-2-bromo-2-chloro-1-fluoroethene trans-1-bromo-1-chloro-2-fluoroethene These compounds possessed strong and characteristic odors. O Alkynes Alkenes O Alkanes Arenesarrow_forward
- What is the possible products for following compound with one equivalent of HBr? 3,4 dimethyl-hepta-2,4-diene a) 3 bromo-4,5-dimethyl hepta-3-ene and 4- bromo-3,4-dimethyl hepta-2- ene b) 3 bromo-3,4-dimethyl hepta-3-ene and 4- bromo-3,4-dimethyl hepta-3- ene c) 3 bromo-4,5-dimethyl hepta-2-ene and 3- bromo-3,4-dimethyl hepta-2- ene d) 3 bromo-4,5-dimethyl hepta-3-ene and 4- bromo-3,4-dimethyl hepta-3- ene e) 3 bromo-4,5-dimethyl hepta-3-ene and 2- bromo-3,4-dimethyl hepta-2- ene O a. e O b. b O c.d O d. c O e. aarrow_forward7. Reaction product of 3-methyl-butene with hydrochloric acid:a) 2-methyl-3-chlorobutaneb) 3-methyl-3-chlorobutanec) 2-chloro-3-methyl butaned) 2-chloro-2-methyl butane 8. What type of reaction takes place when CO2 + H2O and energy are obtained from 3-methyl butyne?a) Hydrogenationb) Halogenationc) Partial oxidationd) Total oxidationarrow_forwardThe two alkenes below react with HI at different rates. CH3CH=CHCH3 and CH₂=CH₂ Draw the structural formula of the MAJOR product formed by the alkene having the HIGHER reaction rate. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. TAYY [ ] در ChemDoodle Ⓡarrow_forward
- What are the starting reactants for the following reaction? 4 X = cyclohexa-1,3-diene; Y = ethyl acrylate X = cyclopenta-1,3-diene; Y = acrylaldehyde X = cyclohexa-1,3-ene; Y = ethyl acrylate X = hexa-1,3-diene; Y = ethyl propiolate CO₂C₂H5arrow_forwardName the alkenes below. Use ONLY E/Z designators to indicate stereochemistry. H3C H Ja H CH₂-C H CH3 CH₂CH3arrow_forwardwhat are the structures of the five alkenes that can produce 3-bromo-3-methylhexane on reaction with HBr 2-ethyl-pentene (E)-3-methylhex-2-ene (Z)-3-methylhex-2-ene (E)-3-methylhex-3-ene (Z)-3-methylhex-3-enearrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
